How to ensure safe and appropriate medication management
Nurses, social educators and pharmacists have reached a consensus on 77 standards for best practice in medication management in the nursing and care service.
Background: Healthcare personnel can have different understandings of what constitutes a safe and appropriate medication management process, from requisition of the medication until it is dispensed to the patient. This can lead to medication management errors. Best practice standards therefore need to be developed for medication management.
Objective: The objective of the study was to illuminate various practical challenges and risk areas in medication management in nursing homes and the community nursing service. We also wanted to build a consensus for detailed solutions to address these challenges and risk areas.
Method: We used the Delphi method and carried out three rounds of Questback questionnaires to collect data. Respondents were asked about the extent to which they agreed with statements relating to medication management using a five-point Likert scale. IBM SPSS was used to analyse the quantitative data. In order to calculate the degree of consensus, we used descriptive statistics with an interquartile range (IQR) <1 as a threshold for consensus. We also collected qualitative data from open-ended questions in the questionnaires. These data were analysed using the Framework method.
Results: The study encompassed 54, 46 and 43 participants in the three rounds respectively, made up of nurses and social educators from 17 municipalities and regulatory pharmacists from a pharmacy in the region. We established a consensus for 77 standards for best practice in medication management.
Conclusion: The consensus-based standards for best practice in medication management in the nursing and care service that were developed in this study may represent an important contribution to safeguarding quality in medication management.
Medication management is an important part of the health care provided to users of municipal nursing and care services. It is a sub-process in the overall treatment of patients and is subject to requirements for professional accountability (1). Medication management is a challenging task. The form, strength and efficacy of medications vary, and they are often prescribed for patients with more than one illness, known as comorbidity (2).
Medication management
Medication management involves any medicine-related task performed from the point the medicine is prescribed or requisitioned until it is dispensed to the patient or discarded (3). Medication management is a complex process that includes preparation, checks, administering to patients, observation and documenting reactions and side effects, as well as reporting any deviations (4).
The current regulations on medication management do not provide detailed instructions on how to address various practical challenges (3). A circular to the Norwegian regulations on medication management (1) gives advice and practical examples intended to maintain good routines and safeguard quality in medication management. However, there is evidence that healthcare personnel still have different understandings of what constitutes safe and appropriate medication management (5), and this can lead to medication management errors from the requisitioning of the medication until it is dispensed to the patient (6).
Medication management errors
Bielecki (4) categorises medication management errors as a breach of one of the ‘7 Rights’. The ‘7 Rights’ entail the right patient receiving the right medication at the right time, in the right form, and in the right way with the right strength or dose.
Medication management errors are defined by Shawahna et al. (6) as deviations from that prescribed in the patient’s medical records, deviations from the manufacturer’s recommendations for storage, expiry date, preparation and administration or deviations from relevant institutional policies. More than 80 per cent of the errors are linked to assembling, preparing and dispensing medications to patients (7).
Medication management errors can be caused by outdated practices, poor routines, the absence of procedures or skills, carelessness or lack of knowledge (8). Simonsen et al. (7) refer to surprising weaknesses in basic knowledge on medication management among nurses in the specialist health service and primary health care. These weaknesses represent a significant potential for error.
Responsibilities in medication management
Healthcare service managers are responsible for development, implementation and compliance in relation to procedures and instructions for medication management. They must also ensure that healthcare personnel have the necessary knowledge and skills within medication management (1). Nevertheless, many studies, both national and international, identify a major need to improve medication management routines (9, 10) and internal controls (11).
Safety procedures are not always possible to carry out in practice, and there is a mismatch between medication competence, work tasks and staffing (11). Healthcare personnel are often interrupted when preparing and administering medication and when monitoring patients’ reactions. The employer’s expectations within medication management are also unclear sometimes (9). Improving competence and dealing with deviations are vital to reducing the risk of errors (7, 11–14).
Pharmaceutical advice and medication audits can reduce the risk of errors in medication management, but Circular IS-7/2015 does not clarify how the advisory aspect should work in practice (10). Role clarification or clarifying responsibilities, establishing interdisciplinary teams and increased knowledge of medication management are among the measures covered.
According to the report on appropriate medication use for elderly patients/residents in nursing homes and in the community nursing service (15), the measures can contribute to optimum use of medicines in primary health care. However, the report has a limited focus on the practicalities entailed in the actual handling of medications in the nursing and care service.
An updated and harmonised list of medications used should always accompany patients when the level of care is changed (3). The literature shows that the manual exchange of information on medications can represent a threat to patient safety (16). An electronic list of medications is a recognised way of safeguarding the quality of information on medications and increasing patient safety (17), but there is still scope for improvement in terms of the electronic exchange of information in Norway (18).
Objective of the study
The objective of the study was to present the practical challenges of medication management in the nursing and care service, and to establish a consensus on how to address these challenges.
Method
The Delphi method involves a consensus processes and associated questionnaire development. This method is a group communication process aimed at conducting detailed surveys and discussions on a particular topic with a view to reaching a consensus (19). The method has been used in a number of surveys, both nationally and internationally (6, 20).
Typical surveys seek to examine ‘what is’, while the Delphi method tries to establish ‘what should or may be’. The Delphi method entails several rounds of questionnaires (19). Researchers collect and analyse data from a questionnaire, and then create a new questionnaire, which is sent to the same respondent group, normally accompanied by the results from the previous round.
The questionnaires are answered anonymously and the respondents do not meet each other, thus preventing group influence during the process. Selecting questions and which indicators should be considered can be a challenge for researchers.
Consensus achieved through this method entails agreement or concurrence in opinions and attitudes between qualified experts within a defined field. Clear criteria should therefore be drawn up for selecting respondents, often referred to as the group or panel of experts (21).
Sample
First, we retrieved the e-mail addresses of chief executives and heads of department from the websites of 26 local authorities in one county. They were all sent an information letter about the study and permission was requested to conduct the survey. Those we contacted were also asked to provide the names and e-mail addresses of charge nurses or healthcare service managers at all relevant municipal services, such as nursing homes, sheltered housing and the community nursing service.
We received 17 positive replies, and subsequently sent letters to the healthcare service managers and charge nurses in the 17 local authorities. The letter asked the recipients to convey information about the study to all nurses and social educators at their institution.
We requested permission to conduct the study among staff at a pharmacy with a regulatory role within pharmaceutics in the nursing and care sector. We collected 183 e-mail addresses of nurses, social educators, pharmacists and department heads.
Ethical considerations
The study was approved by the Norwegian Centre for Research Data, project number 34428. Participation in the study was voluntary. The respondents were informed that they could reserve the right not to receive the questionnaires and withdraw from the study at any time. The data were stored in accordance with research ethics guidelines and the Declaration of Helsinki (22).
Preparing the questionnaire and data collection
The starting point for the questionnaire was a list of the 161 most common medication management problems in nursing homes and in the community nursing service. The list was based on regulatory reports covering more than half of the municipalities in one county in the period 2008–2012. We further refined this list and discussed it with the reference group, which consisted of a researcher and a manager from the municipal nursing and care service.
We formulated 65 statements relating to medication management, which we used to create the questionnaire. Twenty-seven of these statements were used in round one, 21 in round two and 17 in round three. In addition to the initial statements, we devised new statements as a result of the analysis of comments received in rounds one (28 statements) and two (15 statements).
The respondents gave responses to 27, 49 and 32 statements in rounds one, two and three respectively. In the statements, we chose to use both ‘should’ and ‘must’, which is in line with the wording of the regulations on medication management and the associated circular. The respondents were asked to indicate the extent to which they agreed with the statements using a five-point Likert scale with the following values: ‘Completely agree’, ‘Partly agree’, ‘Neither agree nor disagree (neutral)’, ‘Partly disagree’ and ‘Completely disagree’. Alternatively, they could select ‘Don’t know’.
In the first questionnaire round, respondents could comment on each statement, while in the second and third rounds we asked for comments on statements to be assembled thematically. The questionnaire also included questions about work experience, education and function in the organisation. We used the Questback program to design and distribute the questionnaire.
We first conducted a pilot test of the questionnaire in one of the municipalities. The questionnaire was sent to six nurses, and four responded. The respondents were asked to assess whether the statements were understandable and whether potential difficulties could arise when answering the questionnaire.
No relevant issues were identified in the pilot test, and only small, linguistic adjustments were made after consulting the reference group. We conducted the survey in three rounds in the period November 2013 to May 2014. In all three rounds, we sent reminders after one week of sending the first e-mail, and another after two weeks.
Data analysis
Quantitative analysis
We produced descriptive statistics using the analysis tool IBM SPSS Statistics 22.
Since the data were not normally distributed, we used the Mann-Whitney U test (non-parametric test) (23) to compare the responses from different respondent groups. P-value <0.05 was considered to be statistically significant.
Different definitions of consensus have been used in various Delphi studies (24). In our study, we used the interquartile range (IQR) to describe consensus:
- A consensus was considered to be reached in statements with IQR = 0.00.
- No consensus was considered to be reached in statements with IQR > 1.00.
Using the IQR criterion does not allow for distinctions between the different degrees of consensus for statements with IQR = 1.00. The frequency distribution of responses to a statement can be bimodal, meaning that the IQR is greater than 1.00, despite 60 per cent of respondents generally agreeing with the statement. We therefore developed an additional criterion:
- A consensus was considered to be reached in statements with IQR = 1.00 which received generally positive responses (‘completely agree’ and ‘partly agree’) from more than 60 per cent of respondents.
We excluded non-responses and ‘Don’t know’ responses from the analysis.
Qualitative analysis
Respondents’ comments on statements in the questionnaire formed the basis for the qualitative analysis. The comments were entered in Excel and analysed using the framework approach (25, 26). This method enables the researcher to perform a systematic qualitative data analysis and frame the ongoing data collection (25, 26).
The volume of data in our study was relatively small. We therefore used a simplified framework analysis (27). We interpreted the comments after sorting them into categories. The aim of this analysis was also to identify where statements needed to be clarified or reformulated in the next round.
As a result, we changed some statements and added more follow-up statements. Special attention was paid to statements with IQR = 1.00. Here we searched for findings that could undermine the consensus or non-consensus.
Results
We invited a total of 183 nurses, social educators and pharmacists to participate in the study. Eighty-one of these were department heads in municipal nursing and care services. In the first round (n = 183) we received 54 responses (30 per cent). Some of the respondents exercised their right not to receive questionnaires. The number of questionnaires sent out in the second round was therefore 169, and 164 in the third round.
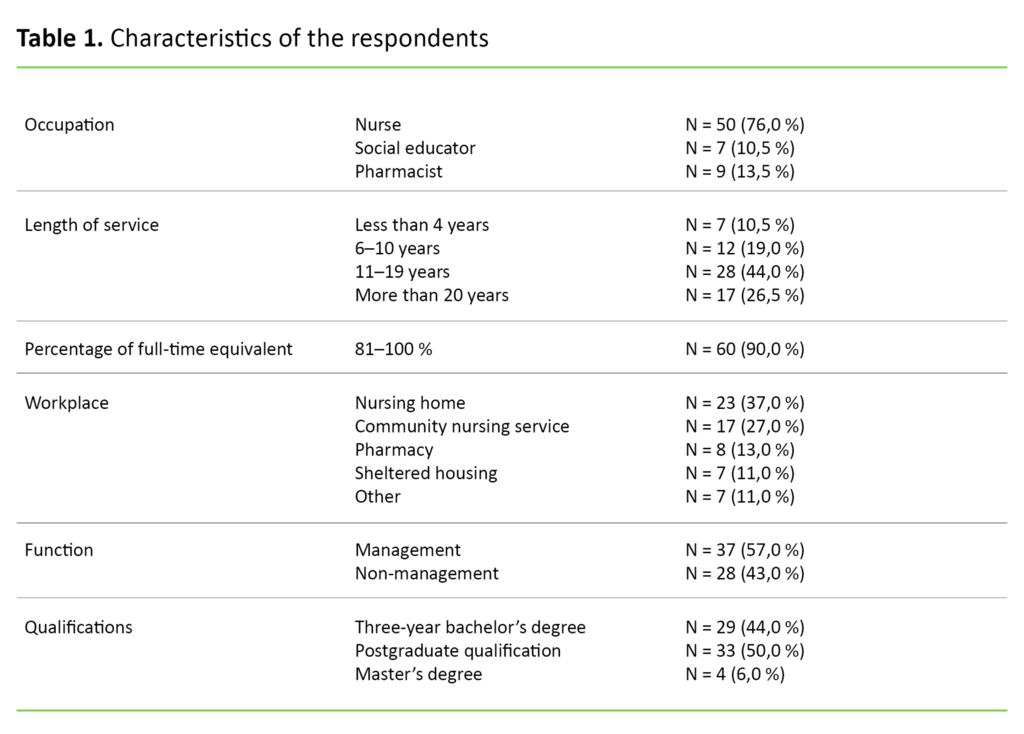
In the second round, we received 46 responses (27 per cent) and in the third round the figure was 43 (26 per cent). In total, 23 respondents answered in all three rounds. Table 1 shows the composition of the respondent group in all rounds. A consensus was reached in the study for 77 standards in medication management (Table 2).
Link to Table 2. Consensus-based standards for best practice in medication management
The response rate in the study was low, with 26–30 per cent, and somewhat lower among respondents without managerial responsibility. A total of 57 per cent of respondents in all three rounds had managerial responsibility. The results did not show significant differences across occupational backgrounds.
Statistically significant differences were found in the study between nurses and pharmacists for nine consensus statements (p-value = 0.001-0.0034). These differences were particularly linked to statements regarding agreements with pharmacies, expiry dates and cross-checks. Nurses and social educators responded differently to four consensus statements concerning loans of emergency supplies, cross-checks and medication storage procedures (p-value = 0.015-0.037).
Statistically significant differences were also found between healthcare personnel with and without managerial responsibility for two statements (p-value = 0.01). The differences between the two groups were linked to statements concerning loans of emergency supplies and cross-checks.
Many statements for which a consensus was not reached also related to cross-checks. Storage of the patient’s private medications in medical supply rooms was another area of non-consensus. Many of the non-consensus statements were reworded or refined, which led to a consensus being reached in the subsequent data collection round.
Discussion
As far as we are aware, this is the first Norwegian Delphi study aimed at developing universal interdisciplinary medication management standards based on Norway’s regulations on medication management. Many studies show that there is a need to improve medication management practices (7, 11–13).
Consensus on medication management standards, which we achieved in this study, may represent an important tool in establishing uniform procedures and serve as a supplement to the medication management regulations for healthcare personnel devising local procedures and routine descriptions.
We achieved a consensus on 77 standards relating to all stages of medication management. We call particular attention to four areas for further discussion: competence, cross-checks, pharmaceutical advice and the exchange of information.
Competence
More than 80 per cent of medication management errors are linked to assembling, preparing and dispensing medications to patients, and can be prevented through increased knowledge and good practices (7). There is often a mismatch between medication management competence, work tasks and staffing (11).
The study does not provide an answer to how safety procedures should be carried out, but specifies that the organisation should devise a training plan that enables permanent employees and agency workers to develop and maintain pharmaceutical competence. We also suggest the content of such a training plan.
Our experiences show that nurses and social educators are rarely given the opportunity to participate in medication management courses, unlike healthcare workers. In many organisations, staff with limited authority in medication management attend refresher courses on this subject every two or three years.
It could be argued that staff groups who have a bachelor’s degree in health and social care studies also need regular updates in medication management. Errors are often caused by actions based on old habits, poor routines or the absence of procedures (8). Participation in the study may therefore have prompted respondents to reflect on current practices.
Healthcare service managers are responsible for ensuring that healthcare personnel possess the necessary knowledge and skills within medication management (1). Dilles et al. (9) emphasise the importance of managers explaining the expectations they have of employees. Healthcare service managers are also responsible for ensuring that routines are developed and monitored. This includes procedures for risk assessment, evaluation, controls and dealing with deviations. Staff throughout the organisation should be familiar with the procedures (3).
In the study, 57 per cent of respondents had managerial responsibility, which could be a crucial factor in implementing the study results in daily practice. The somewhat higher response rates among managers may be due to the fact that it is the manager of a service that is responsible for quality assurance and for updating local guidelines (3).
The list of consensus-based standards in medication management can serve as an aid in defining what should be treated as a deviation, and can be used by healthcare personnel to update guidelines (Table 2). These standards can also help clarify the employer’s expectations in terms of medication management.
Cross-checks
Study respondents put forward recommendations on what is best practice for adhering to the ‘7 Rights’ (4). Medication management errors also include deviations from that prescribed in the patient’s medical records, the manufacturer’s preparation, recommendations and administrative instructions and deviations from relevant institutional procedures (6).
The study has focused on all aspects of the medication management chain, including those taking place in the medical supply room before the medication is administered to the patient. An important measure for preventing errors is cross-checks. The statutory framework does not clarify the use of this measure and views it as a part of healthcare service managers’ risk assessments.
The study identified a consensus for cross-checking medications in pre-prepared dosette boxes, which entails a check by a healthcare professional other than the one who prepared the dosette box. This cross-check should also be a defined task for nurses and social educators.
Statements concerning self-cross-checks did not achieve a consensus, and nurses and pharmacists gave different responses to some of the statements about cross-checks. This can be interpreted to mean that the respondents believe it is important for another qualified employee to perform a quality assurance check.
Pharmaceutical advice
The findings in the study correspond to the measures suggested in the report on appropriate medication use for elderly patients/residents in nursing homes and in the community nursing service (15). Clear responsibilities and role clarification, interdisciplinary teams and a focus on increased competence are all areas that are highlighted in the study.
Employees in the municipal nursing and care service face a variety of barriers to appropriate medication management in relation to organisation, interdisciplinary cooperation and the interaction with patients and their families (9). Research shows that pharmaceutical advice and medication audits can reduce the risk of errors in administering medication (10).
The findings from our study can improve the understanding of the role that pharmaceutical advisors or pharmacists can play in the nursing and care service. Action plans drawn up on the basis of regulatory reports should be made known to staff throughout the organisation, including doctors and pharmacists, and used actively to improve routines. This point achieved a consensus in the study.
Medication reviews are now governed by regulation (3), but even in this study – which was carried out before the regulation was introduced – we achieved a consensus for medication review routines to be established as part of the quality assurance process.
Exchange of information
The literature shows that the manual exchange of information on medications can represent a threat to patient safety (16). Electronic medication lists are recommended for safeguarding quality in the information transferred between service levels (17).
Our findings suggest a need to improve routines surrounding the transfer of responsibility and information on medications between service levels (18), something that the regulations on medication management do not cover in detail. These routines can be added at the system level through electronic exchanges of medication information, but can also be applied in the day-to-day service at an individual level between the patient (and their family) and the professional practitioner.
The service must have routines to ensure that the patient receives the right dose of medicine at the right time and in the right way. This information must be documented in the patient’s medical records (1). The regulations do not specify requirements for documentation in medication management.
We achieved a consensus in the study in relation to who should document medication-related data and what practices should be adopted. Further studies could elaborate on and examine this work in more detail.
Strengths and weaknesses of the study
A strength of the study is the complex use of the Delphi consensus process. Combining qualitative and quantitative analyses, using initial statements and respondents’ comments, enabled us to portray interprofessional ownership of developed standards.
The fact that the respondent group was not exactly the same in each round was a weakness of the study. In addition, the response rate of 26–30 per cent was low. It may be that those who participated are particularly interested in the topic of the study. Their answers may therefore reflect their interest and insight.
The questionnaires were quite extensive, which may have deterred participants from responding to rounds two and three. The total number of respondents was 66, but only 23 participated in all three rounds. However, the majority of earlier Delphi studies have had between 15 and 20 respondents (19). The scope of the dataset can therefore be considered satisfactory.
Although the data were collected in 2013–2014, current medication management practices remain largely unchanged, and we therefore consider our findings to still be pertinent. Further research in other municipalities is necessary to verify the consensus standards that emerged in the study. The barriers to and challenges of implementing standards also need to be clarified.
Conclusion
The study highlights selected risk areas for medication management and the need for a more uniform understanding of safe and appropriate medication management. Consensus-based standards for best practice in medication management, as developed in the study, can play an important role in safeguarding quality in this area.
The list of 77 consensus-based standards from the study can be used to update local guidelines.
References
1. Helsedirektoratet.. Legemiddelhåndteringsforskriften med kommentarer. Oslo: Helsedirektoratet; 2015. Rundskriv IS-7/2015. Available at: https://helsedirektoratet.no/Lists/Publikasjoner/Attachments/942/Rundskriv%20Legemiddelhåndteringsforskriften%20IS-7%202015.pdf(downloaded 29.06.2016).
2. Dahl AA, Grov EK. Komorbiditet i somatikk og psykiatri. Oslo: Cappelen Damm Akademisk; 2014.
3. Helse- og omsorgsdepartementet. Forskrift 3. april 2008 nr. 320 om legemiddelhåndtering for virksomheter og helsepersonell som yter helsehjelp. Available at: https://lovdata.no/dokument/SF/forskrift/2008-04-03-320(downloaded 13.12.2017).
4. Bielecki T, Børdahl B. Legemiddelhåndtering. Oslo: Gyldendal Akademisk; 2014.
5. Fagforbundet. Legemiddelhåndtering – forståelse av forskriften. Oslo: Fagforbundet; 2016. Available at: https://www.fagforbundet.no/yrke/Helsefagarbeider/a/5828/legemiddelhandtering-forstaelse-av-forskriften/(downloaded 13.12.2017 ).
6. Shawahna R, Masri D, Al-Gharabeh R, Deek R, Al-Thayba, Halaweh M. Medication administration errors from a nursing viewpoint: a formal consensus of definition and scenarios using a Delphi technique. Journal of Clinical Nursing. 2016;25(3–4):412–23.
7. Simonsen BO, Johansson I, Daehlin GK, Osvik LM, Farup PG. Medication knowledge, certainty, and risk of errors in health care: a cross-sectional study. BMC Health Services Research. 2011;11:175.
8. Nilsen MK, Sagmo LAB. Multidose i sykehjem. Norsk Farmaceutisk Tidsskrift. 2012;(4):14–8.
9. Dilles T, Elseviers MM, Van Rompaey B, Van Bortel LM, Stichele RRV. Barriers for nurses to safe medication management in nursing homes. Journal of Nursing Scholarship. 2011;43(2):171–80.
10. Fauskanger HT, Irgens MMB. Sykehjem trenger bedre kunnskap om legemidler. Sykepleien; 2010. Available at: https://sykepleien.no/2010/08/sykehjem-trenger-bedre-kunnskap-om-legemidler(downloaded 05.11.2018).
11. Storli M, Ingebrigtsen O, Nakrem S, Elstad TA. Sikkerhetstiltak for legemidler i sykehjem. Sykepleien Forskning; 2016. Available at: https://sykepleien.no/forskning/2016/12/sikkerhetstiltak-legemidler-i-sykehjem-er-ikke-gode-nok(downloaded 05.11.2018).
12. Kirkevold Ø, Engedal KA. Legemiddelhåndtering i norske sykehjem med søkelyset på knusing av medikamenter. Sykepleien Forskning. 2010;5(1):16–25. Available at: https://sykepleien.no/forskning/2010/03/legemiddelhandtering-i-norske-sykehjem-med-fokus-pa-knusing-av-medikamenter(downloaded 05.11.2018).
13. Wannebo W, Sagmo L. Stort behov for mer kunnskap om legemidler blant sykepleiere i sykehjem. Sykepleien Forskning. 2013;8(1):26–34. Available at: https://sykepleien.no/forskning/2013/01/stort-behov-mer-kunnskap-om-legemidler-blant-sykepleiere-i-sykehjem(downloaded 05.11.2018).
14. Simonsen BØ. Evaluering og utvikling av legemiddelkompetansen hos autoriserte sykepleiere. Norsk Farmaceutisk Tidsskrift. 2017;(2):34–36.
15. Helsedirektoratet. Riktig legemiddelbruk til eldre pasienter/beboere på sykehjem og i hjemmesykepleien. Oslo: Helsedirektoratet; 2011. Report IS-1887.
16. Brattheim B, Hellesø R, Melby L. Elektronisk meldingsutveksling ved utskriving av pasienter fra sykehus til kommune. Sykepleien Forskning. 2016;11(1):26–33. Available at: https://sykepleien.no/forskning/2016/02/elektronisk-meldingsutveksling-ved-utskriving-av-pasienter-fra-sykehus(downloaded 05.11.2018).
17. Lyngstad M, Melby L, Grimsmo A, Hellesø R. Toward increased patient safety? Electronic communication of medication information between nurses in home health care and general practitioners. Home Health Care Management & Practice. 2013;25(5):203–11.
18. Borgen K, Melby L, Hellesø R, Steinsbeck A. Elektronisk meldingsutveksling mellom hjemmetjenestene og fastleger. Sykepleien Forskning. 2015;10(1):42–8. Available at: https://sykepleien.no/forskning/2015/02/forventninger-og-erfaringer-med-elektronisk-meldingsutveksling-blant(downloaded 05.11.2018).
19. Hsu CC, Sandford BA. The Delphi technique: Making sense of consensus. Practical Assessment, Research & Evaluation. 2007;12:(10):1–8.
20. Rognstad S, Brekke M, Fetveit A, Spigset O, Wyller T, Straand J. The Norwegian General Practice (NORGEP) criteria for assessing potentially inappropriate prescriptions to elderly patients. A modified Delphi study. Scand J Prim Health Care. 2009;27(3):153–9.
21. Saunes IS, Deilkås E, Kristoffersen M, Lyngstad M, Nyen B. Formaliserte konsensusprosesser, veiledning ved vanskelige valg? Oslo: Kunnskapssenteret. 2009.
22. Helsinkideklarasjonen. WMA Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects. 2013. Tilgjengelig fra: http://legeforeningen.no/PageFiles/175539/Declaration%20of%20Helsinki-English.pdf (downloaded 13.12.2017).
23. Aalen OO, Veierød MB, Frigessi A, Moger TA, Scheel I, Skovlund E. Statistiske metoder i medisin og helsefag. Oslo: Gyldendal Akademisk; 2006.
24. Rayens MK, Hahn EJ. Building consensus using the policy Delphi method. Policy, Politics, & Nursing Practice. 2000;1(4):308–15.
25. Gale KN, Heath G, Cameron E, Rashid S, Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Medical Research Methodology. 2013;13:117.
26. Ritchie J, Spencer L, O'Connor W. Carrying out qualitative analysis. I: Ritchie J, Lewis J, red. Qualitative research practice: A guide for social science students and researchers. London: Sage Publications; 2003.
27. Pope C, Ziebland S, Mays N. Analysing qualitative data. Qualitative research in health care. BMJ. 2000;320:114–6.










Comments