Characteristics of nursing home residents with late-stage dementia and pain
Residents with pain suffered from several health issues and presented with physical as well as mental symptoms. Healthcare personnel can help by providing more effective and appropriate treatment and nursing care.
Background: It has been estimated that more than 50 percent of nursing home residents with late-stage dementia experience pain, but there is little research into the characteristics of these residents. Earlier research on the clinical and demographic characteristics of people with pain is often based on cognitively healthy adults. We therefore need knowledge about the characteristics of elderly nursing home residents who are living with late-stage dementia and pain, as this will shed light on these people’s complex morbidity and needs. Such knowledge may help healthcare personnel develop the observational and assessment skills required to identify intervention needs.
Objective: The objective was to survey the prevalence of pain amongst nursing home residents with late-stage dementia. We also wanted to identify the characteristics of nursing home residents with late-stage dementia and pain by comparing the clinical and demographic characteristics of a group of residents with pain to those of a group of residents with no pain.
Method: The study is a quantitative descriptive study with a cross-sectional design. A total of 112 nursing home residents from 16 nursing homes took part in the study. Registered nurses at each nursing home used a standardised form to collect demographic data. They also made use of a validated screening instrument to collect clinical data concerning pain, neuropsychiatric and depressive symptoms, agitation, personal activities of daily living, somatic health and quality of life. Data concerning the use of analgesics and psycholeptics were obtained from the residents’ medication lists.
Results: The study suggested that two thirds of the sample were living with pain. The mean score for neuropsychiatric symptoms and quality of life was higher among residents who experienced pain (a higher score indicates lower quality of life) compared to residents who did not experience pain. We also found a link between a higher prevalence of depressive symptoms and experiencing pain and a greater need for assistance with personal activities of daily living.
Conclusion: The study reveals that pain is a frequent problem for elderly nursing home patients with late-stage dementia. Our findings suggest that nursing home residents with late-stage dementia and pain experience more symptoms and a lower quality of life than residents with no pain, which may lead to additional needs for treatment and nursing care.
In the Norwegian population, between 80 000 and 100 000 people are living with dementia, and among those who live in nursing homes, approximately 80 percent suffer from dementia (1). Dementia is characterised by cognitive impairment, which affects the individual’s ability to receive, process and organise information, how they use language and how they communicate.
Dementia is normally categorised in three stages: mild, moderate and late-stage dementia. In individuals with late-stage dementia, the ability to think logically and to make appropriate decisions is normally severely impaired (2). The prevalence of comorbid afflictions like stroke, diabetes, chronic heart failure, Parkinson’s and osteoporosis, with associated symptoms such as pain, is high among elderly people with dementia (3).
People with late-stage dementia often have problems associated with verbal language, manifested by struggling to find the right words or by an impaired ability to understand complicated statements. This is one of the reasons why the assessment and treatment of pain tend to be more complicated among this group of individuals compared to among those without dementia or with a mild degree of the disease.
Earlier research
Studies conducted in Norwegian nursing homes have found that pain is experienced by up to 80 percent (4, 5) of residents who suffer from dementia, yet little research has been conducted into the characteristics of these residents. Such knowledge may help us to identify the consequences of failing to meet the needs of nursing home residents who are living with dementia as well as pain.
The knowledge emanating from such a study may also inform healthcare personnel about observations and assessments that will allow them to identify a need for treatment and nursing care, and shed light on these people’s complex morbidity and needs.
There is extensive research on sex differences associated with pain. There are clinical as well as experimental studies (6), and a wide range of clinical conditions are covered (7). Nevertheless, it is our impression after repeated literature searches that there is little research on sex differences associated with pain among the elderly and/or people with cognitive impairment. Consequently, our references do not include research on samples made up of elderly individuals with cognitive impairment.
In a critical literature review, Fillingim et al. concluded that across various clinical conditions such as cancer, musculoskeletal pain, back pain, abdominal pain and neuropathic pain, and experimental studies, pain appears to occur more frequently among women than among men (7).
The pain epidemiology among the elderly is not well understood, and the prevalence of pain among the elderly varies considerably between studies (8). Some studies refer to an increased prevalence of pain with increasing age, while others report the opposite (9).
The few studies from nursing homes that include residents with dementia suggest that there is a link between pain and clinical characteristics such as neuropsychiatric symptoms (NPS) (10), depressive symptoms (11) and agitation (12), and a greater need for help with activities of daily living (ADL) (13).
However, a systematic literature review points to weak associations between these symptoms and pain among people with dementia (14). Furthermore, pain may also be associated with a reduced quality of life (15), impaired mobility, a greater degree of cognitive failure, the use of psycholeptic medication and physical restraint, and impaired verbal communication (16, 17). However, more knowledge is needed (17).
The objective of the study
We wanted to examine the characteristics of nursing home residents with late-stage dementia and pain. The objective of this study was therefore to compare demographic and clinical characteristics between a group of residents who experience pain (hereafter referred to as ‘the pain group’) and a group of residents who do not experience pain (hereafter referred to as ‘the pain-free group’). The study’s research questions were as follows:
- What is the prevalence of pain among elderly nursing home residents with late-stage dementia?
- Are there any statistically significant differences between residents with and without pain in terms of demographic characteristics (gender and age) and clinical characteristics (ADL, NPS, depressive symptoms, somatic health, quality of life, agitation, use of analgesics and psycholeptics)?
Method
The study is a quantitative, descriptive study with a cross-sectional design. We collected the baseline data from a major study that examined the significance of regular pain assessment on pain scores and the use of analgesics in nursing home residents with late-stage dementia (18).
Participants
In random order, we invited nursing homes in four counties in eastern Norway to participate in the study. Registered nurses at each of the nursing homes identified which residents would be appropriate candidates for participation in a two-step procedure:
- Step 1: The registered nurses included residents who were 65 years of age or older, suffered from late-stage dementia and were unable to self-report pain. The latter was a professional assessment made by the nurses based on their knowledge of the resident. Short-term residents of nursing homes (<4 weeks) were not invited to participate the study.
- Step 2: The registered nurses screened the residents who had been identified at step 1. If the resident experienced clinically significant pain, agitation and/or NPS, the resident was included in the study. Clinically significant pain was defined as a score of ≥5 on the Doloplus-2 scale for assessing pain (19), clinically significant agitation was defined as a score of ≥39 on the Cohen-Mansfield Agitation Inventory (CMAI) (20), while clinically significant NPS was defined as a score of ≥4 (frequency x intensity) for one of the questions in the Neuropsychiatric Inventory Nursing Home Version (NPI-NH) (21).
The registered nurses included a total of 121 residents at step 1, and none of these residents were excluded at step 2. Of the 121 residents who were identified at steps 1 and 2, nine were excluded before data collection commenced: one died, two were removed from the study based on the nurse’s assessment, and one nursing home with six residents withdrew from the study.
Data collection
Data were collected in the period between March 2015 and June 2016. Registered nurses at each of the nursing homes used a standardised form to gather demographic data, including gender and age, and validated screening instruments to collect clinical data about the participating residents. The nurses used assessment tools based on observational to collect data as follows:
- Prevalence of pain was measured on the Doloplus-2 scale of pain assessment (19). The scale consists of ten questions that are scored between 0 and 3, where 0 indicates that the behaviour under assessment is ‘normal’ or ‘absent’, while 3 is the highest behavioural score. Total scores range from 0 to 30. A score of ≥5 indicates pain (19). A pain score of ≥5 was used to split the sample in two: ‘the pain group’ (score ≥5) and the ‘pain-free group’ (score ≤4).
- Depressive symptoms were measured using the Cornell depression scale (22), which consists of 19 questions rated as follows: 0 = not present, 1 = moderate or only periodically present, 2 = severely present, and a = cannot be assessed. The score ratings for individual questions were added up to a total, where a score of ≤4 indicates no depressive episode, 7–11 indicate a potentially mild depressive episode, and ≥12 indicates a moderate to severe depressive episode (22).
- Neuropsychiatric symptoms were measured using NPI-NH (23). If the behaviour or symptom is present, an assessment is recorded of its frequency (ranging from 1 = rarely to 4 = very frequently) and intensity (1 = mild, 2 = moderate, 3 = severe). A sub-score is calculated for each behaviour or symptom (frequency x intensity). The sub-scores for the first ten questions are subsequently added up to form a total score. NPI-NH consists of twelve questions, but ‘Sleep and nighttime behavior disorders’ and ‘Appetite/eating abnormalities’’ were not included in the total score for this study. Scores therefore range from 0 to 120.
- Agitation was measured using CMAI, where 29 agitated behaviours are given a score from 1 = never to 7 = several times an hour (24). Total scores range from 29 to 203.
- Personal activities of daily living (p-ADL) were measured by means of an ADL assessment (25). To describe the individual’s need for assistance, six activities are rated on a scale from 0 = not applicable and 1 = total independence to 5 = total dependency. In this study, scores for each question were added up to get a total score. We used a cut off value of ≥18 (score of ≥3 for each element) to indicate a moderate to high need for assistance.
- Quality of life was measured using QUALID, the quality of life in late-stage dementia scale (26), which consists of eleven statements that are assessed on a five-point scale ranging from 1 = never/rarely to 5 = most of the day/once or day. Total scores range from 11 to 55; the lower the score, the higher the quality of life.
- Somatic health (medical comorbidity) was measured using the General Medical Health Rating (27). The residents’ present and former medical histories, apart from their dementia, are grouped in categories 1 to 4 (1 = excellent, 2 = good, 3 = moderate and 4 = poor).
- Use of analgesics and psycholeptics was established based on anonymous copies of the residents’ medication lists. Regular use of analgesics (ACT codes N02A and N02B), psycholeptics (ATC code N05) and antidepressants (ATC code N06A) is a categorical variable: yes/no. We also included a continuous variable for dose, measured in milligram per day of paracetamol and oral morphine equivalents, where all opioids, irrespective of effective substance and form, are combined under a common designation. This refers to the quantity of morphine that the dose would equal if the morphine was administered in tablet form.
Analysis
All data analyses were conducted in SPSS version 25 (IBM Corp., Armonk, NY, USA). We present continuous variables with mean and standard deviation (SD), while categorical variables are presented with frequency and relative frequency as a percentage. We used an independent samples t-test to compare the pain group with the pain-free group for continuous variables. We used a chi-squared test for categorical variables.
Ethical considerations
The residents who participated in the study suffered from late-stage dementia. We therefore obtained written consent from their closest relatives before the study started. The study was approved by the Regional Committees for Medical and Health Research Ethics (REK south-east, reference number 2014/1431).
Results
A total of 112 nursing home residents from 16 nursing homes participated in the study. The mean age of the full sample was 84, and 70 percent were women. Table 1 provides a further description of the sample. One of the main findings is that according to the Doloplus-2 assessments, two thirds of the sample appear to experience pain. See table 2 for information about the prevalence of pain and the use of analgesics.
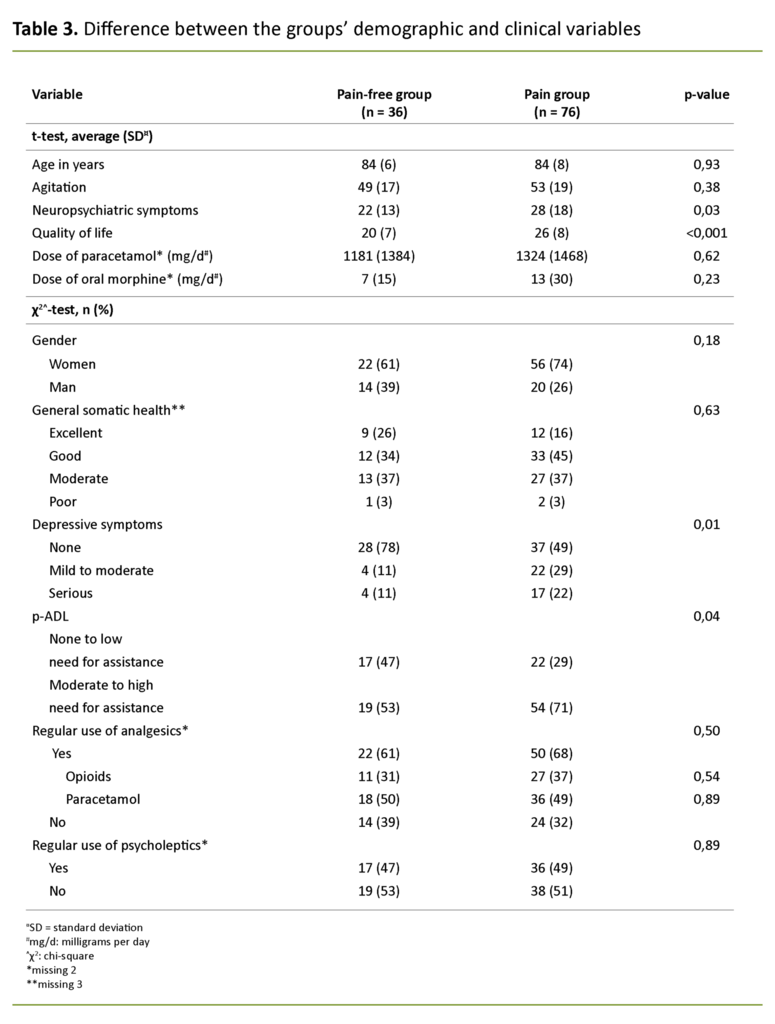
In table 3 we present the difference between the pain group and the pain-free group in terms of clinical and demographic variables. Another main finding is that mean scores for NPS and quality of life (the higher the score, the lower the quality of life) are higher in the pain group than in the pain-free group. We found a link between pain, a higher need for assistance in p-ADL and the prevalence of depressive symptoms.
Discussion
There was a high prevalence of pain among nursing home residents with late-stage dementia. Assessments suggest that two in three residents experience pain. This is not something new, and it matches earlier research from Norwegian nursing homes that has examined the prevalence of pain among residents with late-stage dementia (4).
The results show that one third of the residents who were considered to experience pain, were not receiving analgesics on a regular basis. This does not mean that the residents did not receive any form of pain relief, for PRN (‘as needed’) analgesics may have been administered, or non-pharmacological interventions may have been implemented. It is also interesting to note that two thirds of the residents who were considered to experience pain, did receive analgesics on a regular basis.
Even if a person is receiving analgesics on a regular basis, this does not necessarily mean that the treatment is satisfactory. The fact that so many are considered to be experiencing pain despite regular use of analgesics, may suggest that the pain relief provided is in fact inadequate.
The pain group had a lower quality of life
Our results show that the pain group has a higher mean NPS score compared to the pain-free group, and that there is a link between experiencing pain and a greater need for assistance with p-ADL, as well as a higher prevalence of depressive symptoms. An increased need for assistance with p-ADL is one of the diagnostic criteria for dementia (28) and is one of the most common reasons why people with dementia move into a nursing home (29).
Furthermore, neuropsychiatric and depressive symptoms are frequent among elderly nursing home residents with dementia (30, 31). It is therefore reasonable to assume that many individuals in our sample will be needing assistance with basic p-ADL and/or experience neuropsychiatric and depressive symptoms, irrespective of whether they experience pain.
Nevertheless, we found that the pain group presented with considerably greater physical and mental symptoms and health issues compared to the pain-free group. Our results, supported by earlier research (10, 11, 13), therefore indicate that pain is associated with higher scores for NPS and depressive symptoms as well as a greater need for assistance with p-ADL.
This result impacts on another important finding of this study: that the pain group has a lower quality of life compared to the pain-free group. An earlier study showed that pain has a direct effect on the quality of life of nursing home residents with late-stage dementia, but that the link between pain and quality of life can in part be explained by NPS, depressive symptoms and p-ADL (15).
The fact that the pain group has a lower quality of life compared to the pain-free group may therefore be explained by the observed differences in NPS, depressive symptoms and the need for assistance with p-ADL, rather than simply being caused by pain in itself.
Similar use of medication
There was no difference between the two groups in terms of their regular use of psycholeptics or analgesics, nor in terms of their mean dose of analgesics (mg/day), be it paracetamol or oral morphine equivalents. One possible explanation may be that psycholeptics, particularly antidepressants, are in frequent use among residents with dementia (32).
The same applies for paracetamol (33). Moreover, the use of analgesics such as paracetamol and strong opioids has increased radically among the elderly population in the period between the early 2000s and the present day (33).
No link between pain and age, sex or somatic health
Earlier research has shown that demographic variables such as sex and age impact significantly on pain (7, 9), but in our sample there was no age difference between the pain group and the pain-free group, nor was there a link between pain and sex. We were also unable to find a connection between pain and somatic health.
The majority of residents with pain were in fact considered to be in excellent to good somatic health – they had none or only one unstable somatic condition, used four or fewer medications and presented as healthy or only marginally ailing individuals (27).
Furthermore, we found no statistically significant difference between residents with and without pain in terms of mean agitation score. Results from earlier research on the link between pain and agitation in people with dementia are inconsistent.
Habiger et al. conducted a cluster randomised controlled trial and found that pain treatment led to a lower agitation score, which indicates that pain is the underlying cause of agitation (12).
However, Sampson et al., among others, did not find a link between pain and agitation in their longitudinal study (34). Moreover, agitation is a complex concept which covers a wide range of factors, and it may be that some of the CMAI items are more strongly associated with pain than others (34).
Strengths and limitations
One of the study’s strengths is our use of recognised, validated and reliable assessment tools. However, pain is a subjective experience. It is therefore important to recognise that the use of assessment tools that are based on the observation of behaviours rather than on self-reporting by residents, may be inadequate and may even involve erroneous presentation of the residents’ experience of pain.
However, as of yet there is no other well-established assessment method which appears to be better than the observation approach adopted in this study. Another limitation is that the cut off value (Doloplus-2 ≥5) that was used to divide the sample into a pain group and a pain-free group, has not been validated – it is merely based on the clinical experience of the tool developers.
It is also a limitation that several of the observation-based assessment tools we used include overlapping questions. Overlapping behavioural expressions observed for pain, depressive symptoms and dementia, for example, make it difficult for healthcare personnel to distinguish between these symptoms in clinical practice.
Another weakness is our inability to provide a complete picture of the pain management strategies offered to residents. This is because we have no access to data concerning analgesics administered ‘as needed’, nor any details about non-pharmacological interventions for pain.
It is a strength that the sample’s demographic characteristics are comparable to similar major Norwegian studies in the field. The transferability of the findings of this study to residents in other nursing homes must be considered in the light of the limited sample of only 112 residents at 16 nursing homes in the eastern parts of Norway.
The fact that the data that were analysed stem from only a single point in time, prevents us from commenting on causalities. Although we found statistically significant differences between the pain group and the pain-free group, this does not signify that there is a significant difference between the groups, clinically speaking.
Implications
Pain is a frequent problem among nursing home residents with late-stage dementia. Our study indicates that neuropsychiatric and depressive symptoms increase with pain. Additionally, the residents’ p-ADL function and their quality of life diminish with pain. Recognising pain and administering effective pain relief are therefore important tasks for healthcare personnel because nursing home residents who experience pain suffer from more severe symptoms and health problems compared to pain-free residents
Systematic screening of nursing home residents with late-stage dementia is essential because this can give healthcare personnel guidance with respect to further observations and assessments, and assist with identifying residents who need interventions.
Longitudinal studies of nursing home residents who experience pain will provide important information about how pain, over time, influences the personal activities of daily living, depressive symptoms, NPS and quality of life. This can help with the design of more effective measures while avoiding unnecessary or harmful interventions. In turn, this creates a potential for more appropriate use of scarce resources.
Another interesting area for further research may be to investigate symptom clusters – three or more concurrent symptoms that are related but not necessarily brought on by the same underlying cause.
As indicated by the results of our study, nursing home residents with late-stage dementia and pain do not present with a single symptom or health issue, but complex comorbidity with numerous illnesses and symptoms in addition to an impaired ability to communicate verbally.
Conclusion
Our study suggests that pain in elderly nursing home residents with late-stage dementia is a common problem which influences physical and psychological symptoms as well as other health issues. Our results suggest that nursing home residents with late-stage dementia and pain have a higher mean NPS score compared to pain-free residents, and that there is a link between the prevalence of depressive symptoms and experiencing pain and a need for assistance with p-ADL.
Additionally, our findings indicate that nursing home residents with late-stage dementia and pain have a lower quality of life than pain-free residents. Systematic screening of nursing home residents with late-stage dementia for pain and other symptoms and health issues is therefore an important task for healthcare personnel. It is also an ethical and professional responsibility that can contribute to more effective and appropriate treatment and nursing care.
Hanne Marie Rostad is a co-editor of Sykepleien Forskning. The article was accepted before she became a co-editor.
References
1. Grøholt E, Bøhler L, Hånes H. Folkehelserapporten – helsetilstanden i Norge i 2018. Oslo: Folkehelseinstituttet; 2018.
2. World's Health Organization, Alzheimer’s Disease International. Dementia. A Public Health Priority. Geneva; 2012. Available at: https://apps.who.int/iris/bitstream/handle/10665/75263/9789241564458_eng.pdf;jsessionid=AF546989FADC48F521DA310293FFCFF3?sequence=1 (downloaded 16.10.2019).
3. Bunn F, Burn A-M, Goodman C, Rait G, Norton S, Robinson L, et al. Comorbidity and dementia: a scoping review of the literature. 2014;12(1):192.
4. Torvik K, Kaasa S, Kirkevold Ø, Rustøen T. Pain in patients living in Norwegian nursing homes. Palliative Medicine. 2009;23(1):8–16.
5. Husebø BS, Strand LI, Moe‐Nilssen R, Husebo SB, Ljunggren AE. Pain in older persons with severe dementia. Psychometric properties of the Mobilization-Observation-Behaviour-Intensity-Dementia (MOBID‐2) Pain Scale in a clinical setting. Scandinavian Journal of Caring Sciences. 2010;24(2):380–91.
6. Bartley EJ, Fillingim RB. Sex differences in pain: a brief review of clinical and experimental findings. British Journal of Anaesthesia. 2013;111(1):52–8.
7. Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley J. Sex, gender, and pain: a review of recent clinical and experimental findings. The Journal of Pain. 2009;10(5):447–85.
8. Patel KV, Guralnik JM, Dansie EJ, Turk DC. Prevalence and impact of pain among older adults in the United States: findings from the 2011 National Health and Aging Trends Study. 2013;154(12):2649–57.
9. Jones G, Macfarlane G. Epidemiology of pain in older persons. In: Gibson S, Weiner D, eds. Pain in older persons. Seattle: IASP Press; 2005.
10. Husebø BS, Ballard C, Sandvik R, Nilsen OB, Aarsland D. Efficacy of treating pain to reduce behavioural disturbances in residents of nursing homes with dementia: cluster randomised clinical trial. BMJ. 2011;343:d4065.
11. Erdal A, Flo E, Selbaek G, Aarsland D, Bergh S, Slettebo DD, et al. Associations between pain and depression in nursing home patients at different stages of dementia. Journal of Affective Disorders. 2017;218:8–14.
12. Habiger TF, Flo E, Achterberg WP, Husebo BS. The interactive relationship between pain, psychosis, and agitation in people with dementia: results from a cluster-randomised clinical trial. Behavioural Neurology. 2016; Article ID 7036415. DOI: 10.1155/2016/7036415
13. Cadogan MP, Edelen MO, Lorenz KA, Jones M, Yosef J, Hascall T, et al. The relationship of reported pain severity to perceived effect on function of nursing home residents. The Journals of Gerontology. Series A: Biological Sciences and Medical Sciences. 2008;63(9):969–73.
14. van Dalen-Kok AH, Pieper MJ, de Waal MW, Lukas A, Husebo BS, Achterberg WP. Association between pain, neuropsychiatric symptoms, and physical function in dementia: a systematic review and meta-analysis. BMC Geriatrics. 2015;15(1):49.
15. Rostad HM, Puts MT, Småstuen MC, Grov EK, Utne I, Halvorsrud L. Associations between pain and quality of life in severe dementia: a Norwegian cross-sectional study. Dementia & Geriatric Cognitive Disorders Extra. 2017;7(1):109–21.
16. Miu D, Chan K, Geriatrics. Under-detection of pain in elderly nursing home residents with moderate to severe dementia. Journal of Clinical Gerontology. 2014;5(1):23–7.
17. Takai Y, Yamamoto-Mitani N, Okamoto Y, Koyama K, Honda A. Literature review of pain prevalence among older residents of nursing homes. Pain Management Nursing. 2010;11(4):209–23.
18. Rostad HM, Utne I, Grov EK, Småstuen MC, Puts M, Halvorsrud L. The impact of a pain assessment intervention on pain score and analgesic use in older nursing home residents with severe dementia: A cluster randomised controlled trial. International Journal of Nursing Studies. 2018;84:52–60.
19. Lefebvre-Chapiro S. The DOLOPLUS-2 scale – evaluating pain in the elderly. European Journal of Palliative Care. 2001;8(5):191–4.
20. Husebø BS, Ballard C, Cohen-Mansfield J, Seifert R, Aarsland D. The response of agitated behavior to pain management in persons with dementia. The American Journal of Geriatric Psychiatry. 2014;22(7):708–17.
21. Selbæk G, Kirkevold Ø, Engedal K. The prevalence of psychiatric symptoms and behavioural disturbances and the use of psychotropic drugs in Norwegian nursing homes. International Journal of Geriatric Psychiatry. 2007;22(9):843–9.
22. Alexopoulos GS, Abrams RC, Young RC, Shamoian CA. Cornell scale for depression in dementia. Biological Psychiatry. 1988;23(3):271–84.
23. Cummings JL, Mega M, Gray K, Rosenberg-Thompson S, Carusi DA, Gornbein J. The Neuropsychiatric Inventory comprehensive assessment of psychopathology in dementia. Neurology. 1994;44(12):2308.
24. Cohen-Mansfield J. Conceptualization of agitation: results based on the Cohen-Mansfield agitation inventory and the agitation behavior mapping instrument. International Psychogeriatrics. 1997;8(S3):309–15.
25. Lawton M, Brody E. Assessment of older people: self-maintaining and instrumental activities of daily living. Nursing Research. 1970;19(3):278.
26. Weiner MF, Martin-Cook K, Svetlik DA, Saine K, Foster B, Fontaine C. The quality of life in late-stage dementia (QUALID) scale. Journal of the American Medical Directors Association. 1999;1(3):114–6.
27. Lyketsos CG, Galik E, Steele C, Steinberg M, Rosenblatt A, Warren A, et al. The General Medical Health Rating: a bedside global rating of medical comorbidity in patients with dementia. Journal of the American Geriatrics Society. 1999;47(4):487–91.
28. Direktoratet for e-helse, Verdens helseorganisasjon. ICD-10: Den internasjonale statistiske klassifikasjonen av sykdommer og beslektede helseproblemer [internett]. Oslo: 2019 [updated 12.08.2019; cited 16.10.2019]. Available at: https://ehelse.no/standarder/ikke-standarder/icd-10-den-internasjonale-statistiske-klassifikasjonen-av-sykdommer-og-beslektede-helseproblemer
29. Verbeek H, Meyer G, Challis D, Zabalegui A, Soto ME, Saks K, et al. Inter‐country exploration of factors associated with admission to long‐term institutional dementia care: evidence from the RightTimePlaceCare study. Journal of Advanced Nursing. 2015;71(6):1338–50.
30. Selbæk G, Engedal K, Bergh S. The prevalence and course of neuropsychiatric symptoms in nursing home patients with dementia: a systematic review. Journal of the American Medical Directors Association. 2013;14(3):161–9.
31. Iden KR, Engedal K, Hjorleifsson S, Ruths S. Prevalence of depression among recently admitted long-term care patients in Norwegian nursing homes: associations with diagnostic workup and use of antidepressants. Dementia & Geriatric Cognitive Disorders. 2014;37(3–4):154–62.
32. Helvik A-S, Benth JŠ, Wu B, Engedal K, Selbæk G. Persistent use of psychotropic drugs in nursing home residents in Norway. BMC Geriatrics. 2017;17(1):52.
33. Berg C, ed. Reseptregisteret 2013–2017. Legemiddelstatistikk 2018:2, Oslo: Folkehelseinstituttet; 2018. Available at: https://www.fhi.no/globalassets/dokumenterfiler/rapporter/2018/reseptregisteret-2013_2017-temadel-om-legemidler-og-eldre.pdf (downloaded 16.10.2019).
34. Sampson EL, White N, Lord K, Leurent B, Vickerstaff V, Scott S, et al. Pain, agitation, and behavioural problems in people with dementia admitted to general hospital wards: a longitudinal cohort study. Pain. 2015;156(4):675.












Comments