A survey of healthcare professionals’ experiences with the Paediatric Early Warning Score (PEWS)
PEWS promotes a systematic approach to monitoring and better communication in paediatric departments, but there is a need to follow up and improve guidelines and quality-assurance activities.
Background: The Paediatric Early Warning Score (PEWS) is a scoring tool that was developed in the early 2000s in England and modified for Norwegian conditions in 2011. The tool quantifies the severity of children’s clinical condition, regardless of diagnosis, and identifies children between 0 and 18 years of age who are at risk of clinical deterioration. PEWS is regarded as a useful tool for recognising at-risk patients, but research on user experiences with PEWS is limited.
Purpose: To describe the experiences of nurses and doctors with PEWS. We describe these experiences in terms of whether the tool contributes to a systematic approach to monitoring and better communication, as well as whether the tool is applicable to the user. In addition, we investigate whether PEWS is used according to established guidelines and whether different hospitals or professional groups have different experiences with the tool.
Method: We used a quantitative method with a descriptive cross-sectional design. We developed our own questionnaire to survey the user experiences of 172 healthcare professionals at three Norwegian hospitals, and performed descriptive analyses with non-parametric rank-sum tests (Kruskal-Wallis and Mann-Whitney-Wilcoxon).
Results: There is general agreement among the healthcare professionals that PEWS promotes a systematic approach to monitoring and better communication and that PEWS is a user-friendly tool. The healthcare professionals at one hospital agreed significantly more often that they use PEWS according to established guidelines, as compared with the other hospitals.
Conclusion: The healthcare professionals report positive experiences with PEWS, but the study shows that they use the guidelines in very different ways.
It is the responsibility of paediatric nurses to ‘evaluate acute situations, set priorities and implement measures that help to maintain or restore vital functions’ (1). This requires systematic monitoring of children who are hospitalised. To prevent acute situations, nurses must quality assure the monitoring of sick children so that any deterioration in the child’s clinical condition can be identified at an early stage.
Paediatric Early Warning Score (PEWS)
The Paediatric Early Warning Score (PEWS) is a scoring tool that quantifies the severity of children’s clinical condition, regardless of diagnosis. Monaghan’s version of PEWS, which was developed in the early 2000s in England, was translated into Norwegian, modified and validated in 2011 at Akershus University Hospital.
A growing number of paediatric departments in Norway have implemented the tool, and a joint initiative to implement the tool nationally is currently underway. The tool is based on normal values on clinical parameters according to the child’s age (0–18 years) within three categories: respiration (AB), circulation (C) and behaviour (D). Increasing deviation from normal values for the child’s age raises the score. A total score indicates the patient’s risk of clinical deterioration (2).
Several validation studies of PEWS have been carried out (3–10). Individual studies are summarised in review articles (11, 12), and reports discuss the implementation of PEWS and evaluate the tool (4, 13, 14). Research shows that PEWS improves, to varying degrees, the ability of healthcare professionals across professions to identify early clinical deterioration in sick children.
A survey of employees’ experiences following the implementation of PEWS (13) and a collection of nurses’ feedback on the use of PEWS show that PEWS gives nurses more self-confidence in their work with sick children. They are uncertain, however, whether their own assessment skills are more effective than the tool (14).
Researchers are seeking more knowledge about users’ experiences of the tool’s applicability. If healthcare professionals are to prevent and identify clinical deterioration and improve the outcome of treatment by using PEWS, the healthcare professionals group must focus on continuing to use the tool (15).
Purpose of the study
It is important to evaluate the tool in order to find out whether PEWS identifies symptoms of clinical deterioration in the child’s condition and that it works as intended. One aspect of the evaluation will also be to elicit the experiences of healthcare professionals and to identify areas for potential improvement (16). However, we have not found studies that have investigated this topic in Norway.
Thus, the purpose of this study is to describe healthcare professionals’ experiences with PEWS in three Norwegian hospitals.
We formulated three main research questions:
- Does PEWS promote a systematic approach to the monitoring of sick children?
- Does PEWS help to improve communication?
- How do users perceive the applicability of PEWS?
- Is the tool used according to established guidelines?
We also formulated the following secondary question:
- Do the responses differ across hospitals or professional groups?
Method
Design
We used a quantitative method with a descriptive design, and conducted a cross-sectional study of paediatric departments in three Norwegian hospitals.
Questionnaire
A validated questionnaire that surveys users’ experiences with PEWS was not available, so we developed our own. The questionnaire contains statements about PEWS that are divided into four main topics which we believed to be especially important for quality-assured monitoring of sick children: a systematic approach, communication, applicability and guidelines.
The statements are based on research findings (3–5, 9, 10, 13, 14) and relevant professional literature. The questionnaire was adapted following a pilot study conducted in autumn 2015 in two hospital departments that had recently introduced PEWS. We also based it on feedback from the individuals who promoted the introduction of PEWS in Norway.
The questionnaire begins with three demographic questions. These are followed by 37 different statements about PEWS, which are divided into the aforementioned topics. The response options are on a Likert Scale with the following options: ‘strongly disagree’, ‘slightly disagree’, ‘neither agree nor disagree’, ‘slightly agree’ and ‘strongly agree’. The respondents were asked to rank their perceptions of the statements according to their experience with PEWS.
The sample
We used a non-probability sample recruited according to predefined criteria (17, 18). Our intention was to recruit nurses, doctors, auxiliary nurses and paediatric nurse assistants who work in paediatric departments and use PEWS. To avoid recruiting in an implementation phase and to ensure that the respondents had clinical experience with children in hospital and PEWS, the inclusion criteria were as follows:
- PEWS implemented at least two years ago
- Healthcare professionals must have a minimum of three months’ clinical experience
We contacted three hospitals in different counties, and they agreed to participate. One was a university hospital and two were regional hospitals. The three hospitals together have four medical and surgical wards for children, as well as one paediatric admission unit. We refer to the entire sample as healthcare professionals. For reasons of personal privacy, we combined nine paediatric nurse assistants and auxiliary nurses with the nursing group in the analysis.
Data collection
Data were collected in spring 2016 over a three-week period. We sent information to the management in advance, and we visited the various departments in person. We provided thorough information about the project and established a cooperative relationship with the contact persons, who assumed special responsibility for recruitment and data collection.
The questionnaires were distributed in paper form to the nurses during their shift change and at joint meetings of the doctors. Everyone who satisfied the inclusion criteria at that point in time received an invitation to participate. The respondents gave their consent by answering the questionnaire. To ensure voluntary participation, everyone received a questionnaire together with an envelope so that they could also choose to return an unanswered form.
Altogether 177 of 220 potential respondents returned completed questionnaires, which is a response rate of 80 per cent. The response rate at Hospital A is 86 per cent, at Hospital B 92 per cent and Hospital C 70 per cent. Five questionnaires were later withdrawn from the analysis because the respondents did not satisfy the inclusion criteria.
Analysis
We performed descriptive analyses of the data. We used non-parametric rank-sum tests (Kruskal-Wallis and Mann-Whitney-Wilcoxon) to compare the hospitals and professional groups.
To simplify the data processing, we consolidated the values ‘slightly disagree / strongly disagree’ and ‘slightly agree / strongly agree’ into ‘disagree’ and ‘agree’, respectively. We used IBM SPSS, version 23 for the statistical analyses, and the significance level was set at 5 per cent, corresponding to a p-value of < 0.05.
Missing data
Under demographic data, three respondents (2 per cent) did not state their profession, seven (4 per cent) did not report their experience with children, and eleven (6 per cent) did not state their experience with PEWS. A maximum of 8 per cent did not answer the other statements on the questionnaire.
Approvals
The study has been approved by internal research coordinators at the hospitals and the data protection official for research, the Norwegian Centre for Research Data (NSD). The data have been treated confidentially. We did not find it relevant to seek approval from the Regional Committees for Medical and Health Research Ethics (REC) (19).
Results
The distribution of nurses and doctors is relatively the same at the three hospitals. In total, 75 per cent are nurses and the rest are doctors (Table 1). Over half of the sample have more than five years of experience with hospitalised children. At Hospitals A and B, over one-quarter of the respondents have more than 15 years of experience. Only 12 percent of the total sample have less than one year of experience with PEWS.
Systematic approach
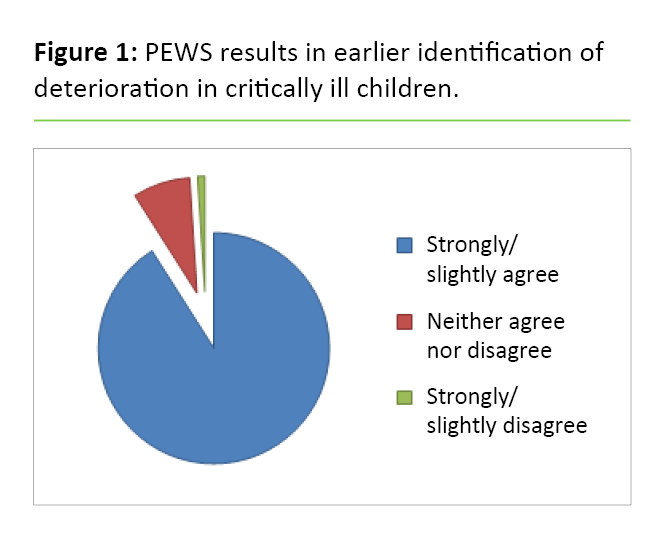
At all three hospitals, over 90 per cent generally agree with the statements that PEWS promotes a systematic approach to the monitoring of sick children (Table 2). A total of 91 per cent agree that PEWS results in earlier identification of clinical deterioration (Figure 1). The results show no significant differences between the hospitals or among the professional groups regarding a systematic approach.
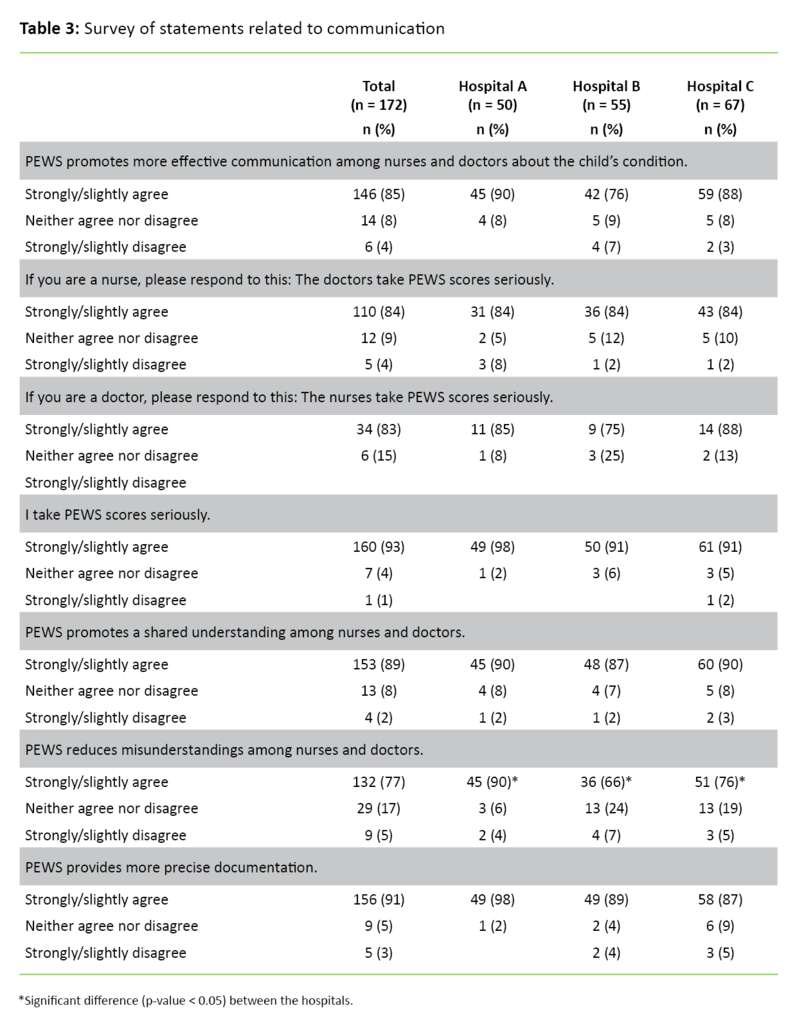
Communication
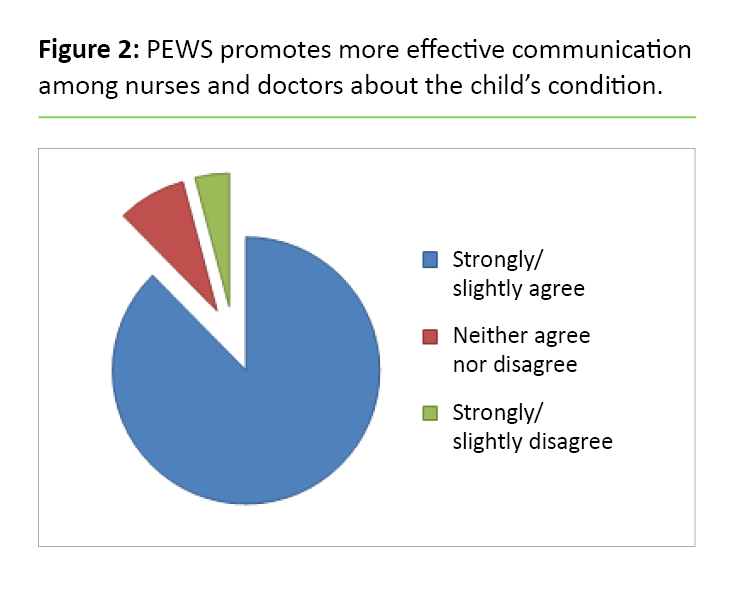
Altogether 85–90 per cent of the sample agree that PEWS promotes more effective communication (Figure 2) and a more shared understanding among the healthcare professionals (Table 3). There is a significant difference between Hospitals A and B (p = 0.01) and Hospitals A and C (p = 0.047) regarding how much the respondents agree that PEWS reduces misunderstandings among healthcare professionals. Healthcare professionals at Hospital A agree more with this statement.
Applicability
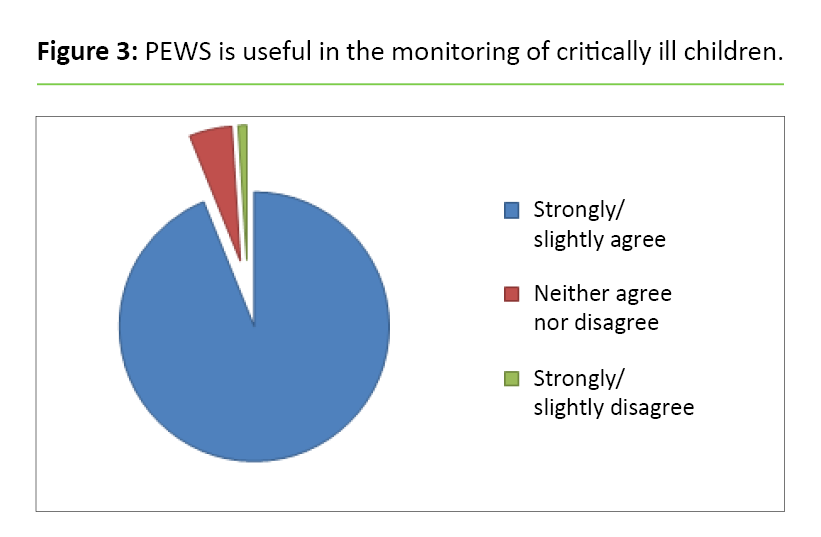
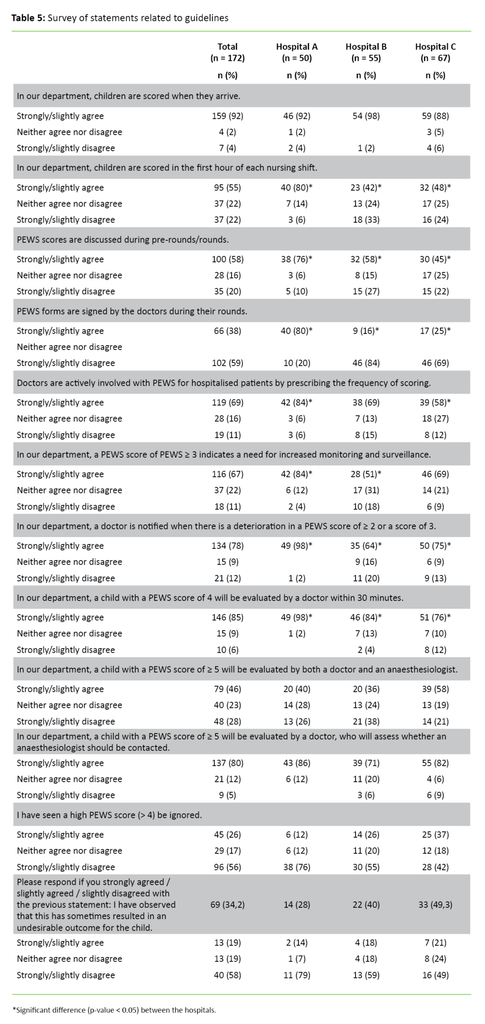
Of all the respondents, 92 per cent agree that PEWS is useful (Figure 3). Altogether 80 per cent state that they have received training in PEWS, although fewer doctors than nurses report this at all the hospitals (A: p = 0.01, B: p = 0.04, C: p = 0.03) (Table 4). In addition, a total of 93 per cent state that they have confidence in the scoring.
Fewer doctors than nurses state that they carry out PEWS scoring (p < 0.001). More of the respondents at Hospital A compared with Hospital B (p = 0.01) agree that PEWS measures whether there is a risk of clinical deterioration in the child’s condition.
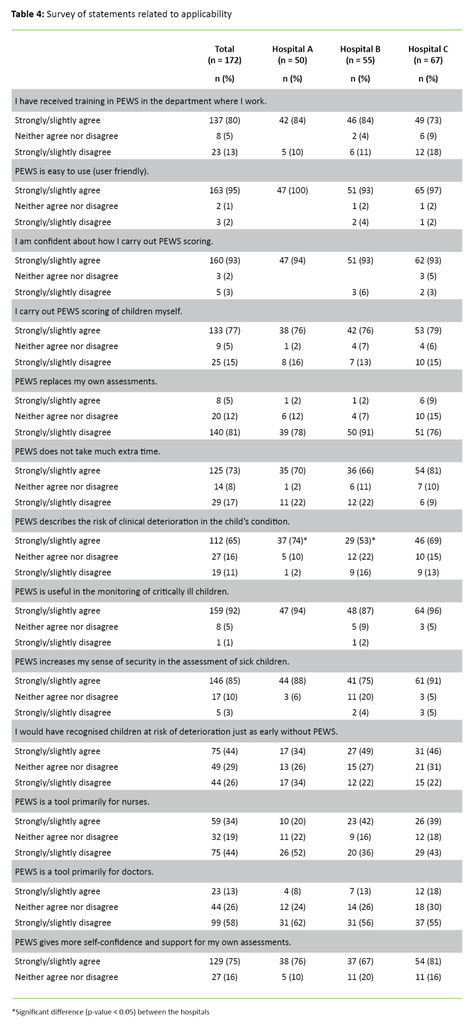
Guidelines
The responses are generally distributed among the response options (Table 5). At Hospital A, more of the respondents agree that the children are scored during the first hour than at Hospitals B and C (A and B: p = 0.01, A and C: p = 0.01). At Hospital A, they also agree that the PEWS form is signed by a doctor during rounds (A and B: p < 0.001. A and C: p < 0.001).
Regarding the question of whether doctors are actively involved with PEWS for hospitalised patients and prescribe the frequency of scoring, more of the respondents at Hospital A than at Hospital C agree with this (p = 0.01).
More respondents at Hospital A than at Hospital B agree that a PEWS score ≥ 3 results in increased monitoring (p = 0.01). In addition, more respondents at Hospital A than at Hospitals B and C agree that a worsening of PEWS by ≥ 2 or a score of 3 leads them to inform a doctor (A and B: p < 0.001, A and C: p = 0.01).
Discussion
It is positive that almost all respondents state that PEWS results in earlier identification of clinical deterioration in sick children and that the tool is easy to use. These findings correspond with previous studies (3–5, 9, 10, 13). If using a systematic tool helps healthcare professionals to recognise symptoms in sick children at an earlier stage, it is an important means of improving the quality assurance of monitoring (20).
Systematic approach
Almost all the respondents agreed that PEWS results in a systematic monitoring of clinical parameters. They also agreed that PEWS leads to a standardisation of the parameters that are used and that the tool ensures continuity in monitoring. When PEWS is used, systematic observations help to identify changes in the patient’s clinical condition.
A systematic approach to changes in the child’s vital signs is crucial for assessing how ill the child is (21). Experience shows that observations and assessments can often be unsystematic and dependent on the individual doctor’s or nurse’s experience and knowledge.
Studies have indicated that routine observations of hospitalised children were carried out in an unsystematic manner and were dependent on the nurse’s perceptions, the doctor’s preference and their clinical specialty. They conclude that the lack of consistency in observing and registering vital signs must be improved because it is critical that a thorough set of observations are recorded for sick children (22). It is therefore useful to have a tool that systematises the monitoring activity.
Almost all respondents agreed that PEWS is a supplement to their daily work which ensures that a minimum of clinical observations are carried out, but that PEWS does not replace their own professional assessments. These findings show that PEWS does not replace either the professional knowledge or the experience that serves as a basis for how healthcare professionals observe the sick child, but that they regard PEWS as a useful aid (2).
Communication
As in previous research (3, 4), healthcare professionals from the hospitals mostly agree that PEWS results in more effective communication and a shared understanding across professional groups, which is especially important when working with sick children, most notably in acute situations (23, 24). It is a strength that many of the respondents confirm that PEWS contributes to this, and other studies also mention improved collaboration and communication as positive reasons to use PEWS (2, 10).
There are some divergent experiences as to whether PEWS reduces misunderstandings among healthcare professionals. A significantly higher percentage of healthcare professionals at Hospital A agree with this statement, compared with Hospitals B and C. More respondents from Hospitals B and C answered ‘neither agree nor disagree’, so we cannot assume that they disagree. It may be that the question is stated unclearly and does not distinguish between ‘whether they have experience with it’ or ‘whether they do not understand the question’.
However, the fact that one-quarter of the entire sample agrees that PEWS reduces misunderstandings is especially important as it pertains to monitoring of the child. Other studies also suggest that misunderstandings related to the patient’s condition are reduced when PEWS is used (10).
Applicability
The respondents confirm that PEWS is easy to use. Many state that they received training in the use of PEWS, but one weakness of the implementation is that not everyone who uses PEWS has been trained in its use. Fewer doctors than nurses have received training, and significantly fewer doctors than nurses carry out the scoring themselves. The explanation for this may be that the scoring has been completed by the time the doctors come to check on the child, and as such it is unnecessary to do the scoring again.
It is a strength that most respondents state that they have confidence in how they conduct the scoring. We assume that more doctors would also have done it themselves if it were necessary. By the same token, it is a weakness that few of the doctors have received PEWS training.
Previous studies suggest that healthcare professionals were concerned that PEWS would be time-consuming and increase their paperwork (13). In our study, less than one-fifth of the respondents believe that PEWS takes a lot of extra time. However, we are surprised that so many still believe that it takes more time. In our view, the observations included in the tool are no different from the daily monitoring that should be recorded.
The advantage of PEWS is that by systematising the monitoring, healthcare professionals are better able to follow the patient’s clinical development over time. In future improvement efforts, it will be important to consider what makes some professionals feel that PEWS is time consuming, even though it can simplify their daily work and is intended to quality assure the monitoring of sick children.
Many of the respondents agree that PEWS is useful and that it increases their sense of security about their assessment of sick children, boosts their self-confidence and supports their own medical assessment. At the same time, relatively few agree that PEWS shows the degree of risk of clinical deterioration, and almost half agree that they would have recognised the risk just as early without PEWS.
One explanation for these findings could be that the statements were not worded clearly, especially because many responded ‘neither agree nor disagree’. However, this may not be the entire explanation, as PEWS received varying feedback from healthcare professionals when the scoring tool was introduced in other countries.
In addition, many believed that they were able to recognise the patient’s risk of clinical deterioration without the tool (14). Despite this, we see both in this study and in previous research that it is viewed as beneficial to use the tool and that the advantage of PEWS is that it gives healthcare professionals greater security, more self-confidence and better support when they assess sick children (2–4, 10, 13).
Guidelines
The findings show that the hospitals have different experiences with the established guidelines for the use of PEWS. Previous studies emphasise that it is important to have a culture that supports the use of PEWS if the tool is to serve its purpose (15). It is a weakness that there is not more agreement among healthcare professionals that the established guidelines must be followed.
Regarding the issue of guidelines, there is a generally significantly higher percentage of agreement among the respondents at Hospital A than at the other hospitals. One possible explanation for the differences may be the length of time that they have been using PEWS in practice. However, this assumption does not correspond with the findings from our study in which Hospitals A and B have both been using PEWS for two and a half years, while Hospital C has been using PEWS for five years.
There is a higher percentage of paediatric nurses and doctors at Hospital A than at the other hospitals, and the possibility that expertise has a bearing on how the guidelines are followed cannot be ruled out. Research also shows that up to 70 per cent of improvement efforts do not maintain the results over time if one does not maintain the focus and view the work as an ongoing process (16). Perhaps Hospital A has been more successful in sustaining motivation within the healthcare professionals group that supports the use of PEWS (15).
The findings also show that over one-third of the respondents have seen a high PEWS score (> 4) be ignored. Altogether 19 per cent of these respondents agree that this resulted in an undesirable outcome for the child, although no further explanation was given.
The respondents at Hospitals B and C agree the least that PEWS is used according to the tool’s established guidelines. At the same time, it is from these two hospitals that most respondents state they have seen a high PEWS score be ignored and that this resulted in an undesirable outcome for the child. The numbers are low, but the findings are serious. Research shows that when clinical deterioration of the child’s condition occurs, it is a major problem when healthcare professionals fail to react appropriately and correctly (25).
Strengths and limitations
The response rate in our study is high, and the analysis shows that few of the responses to the questionnaire lacked data. We chose hospitals where the tool was well established, and we recruited from hospitals in different geographic locations. The participants in the study represent healthcare professionals who use PEWS in their clinical work. The aforementioned measures strengthen the study’s external validity and generalisability (17, 26, 27).
A limitation of the study is that we did not have a validated questionnaire to survey healthcare professionals’ experiences with PEWS. There is a risk that the respondents may have misunderstood the questions, which constitutes a validity threat (17). Prior to this study, we launched measures to minimise this threat by conducting a pilot study, gathering feedback from the driving forces behind PEWS in Norway, and putting a great deal of effort into recruitment.
The fact that the analysis mostly shows similar results among different hospitals is a strength, but a large percentage responded ‘neither agree nor disagree’ to certain statements. This may mean that those statements were not worded clearly, which poses a threat to concept validity. We take this into account when we discuss the results.
None of the authors is affiliated with the hospitals, and thus there are no validity conflicts or bias in this regard (17).
Conclusion
The study shows that the respondents generally agreed that PEWS promotes a systematic approach to monitoring and better communication and that healthcare professionals find the tool to be applicable. The general experiences with the tool seem to be positive.
It is worth noting that healthcare professionals have different experiences with the guidelines introduced for PEWS. The differences are greatest between Hospital A and the other hospitals. The respondents at Hospital A have a considerably higher, and in some cases a significantly higher, level of agreement about the use of the guidelines than the respondents at the other two hospitals.
The findings provide important knowledge about healthcare professionals’ experiences with PEWS, and may thus be important for further quality efforts, with regard both to follow-up and to improvements. The findings indicate the importance of keeping the spotlight on PEWS in the healthcare professionals group and viewing the work with PEWS as an ongoing process in order for the tool to be applicable.
The findings cannot be generalised as a matter of course, but they may nonetheless be useful for everyone who has used or will use the tool in clinical practice since the response rate is high and the sample is relatively large in terms of the number of professionals in Norway who have experience with PEWS. The findings may lead to better follow-up and utilisation of the tool at the hospitals that use PEWS and a better planning phase for implementation of the tool at other health trusts.
Further research
In order to compare the study and evaluate its results, more research is needed on users’ experiences with PEWS. Further research should investigate whether there are reasons that can explain the difference in the use of guidelines. Qualitative studies could help to increase our insight into these issues.
References
1. Høgskolen i Oslo og Akershus. Programplan for mastergradsstudium i barnesykepleie. 2015. Available at: http://www.hioa.no/Studies/HF/Master/Barnesykepleie/Programplan-for-Mastergradsstudium-i-barnesykepleie-2015(downloaded 26.02.2016).
2. Bjerke AT, Moen E, Bråthen A-DB, Schröder J, Solevåg AL, Nakstad B. PedSAFE. Et utdannings- og treningsprogram for helsepersonell som jobber med barn og ungdom. Tidsskrift for barnesykepleiere 2015;1:5–6.
3. Akre M, Finkelstein M, Erickson M, Liu M, Vanderbilt L, Billman G. Sensitivity of the Pediatric Early Warning Score to identify patient deterioration. Pediatrics 2010;125(4):763–9. DOI: 10.1542/peds.2009-0338.
4. Ennis L. Paediatric early warning scores on a children’s ward: a quality improvement initiative. Nurs Child Young People 2014;26(7):25–31. DOI: 10.7748/ncyp.26.7.25.e478.
5. Fuijkschot J, Vernhout B, Lemson J, Draaisma JMT, Loeffen JLCM. Validation of a Paediatric Early Warning Score: first results and implications of usage. Eur J Pediatr 2015;174:15–21. DOI: 10.1007/s00431-014-2357-8.
6. Parshuram C, Duncan H, Joffe A, Farrell C, Lacroix J, Middaugh K, et al. Multicentre validation of the bedside paediatric early warning system score: A severity of illness score to detect evolving critical illness in hospitalised children. Crit Care 2011;15:1–10.
7. Parshuram C, Hutchison J, Middaugh K. Development and initial validation of the bedside paediatric early warning system score. Crit Care 2009;13R135. DOI: 10.1186/cc7998.
8. Skaletzky S, Raszynski A, Totapally B. Validation of a modified pediatric early warning system score: A retrospective case-control study. Clin Pediatr (Phila) 2012;51(5):431–5. DOI: 10.1177/0009922811430342.
9. Solevåg AL, Eggen EH, Schröder J, Nakstad B. Use of a Modified Pediatric Early Warning Score in a Department of Pediatric and Adolescent Medicine. PloS One 2013;8(8):1–6. DOI: 10.1371/journal.pone.0072534.
10. Tucker K, Brewer T, Baker R, Demeritt B, Vossmeyer M. Prospective evaluation of a pediatric inpatient early warning scoring system. J Spec Pediatr Nurs 2009;14(2):79–85. DOI: 10.1111/j.1744-6155.2008.00178.x.
11. Chapman SM, Grocott MP, Franck LS. Systematic review of paediatric alert criteria for identifying hospitalised children at risk of critical deterioration. Intensive Care Med 2010;36(4):600–11. DOI: 10.1007/s00134-009-1715-x.
12. Murray JS, Williams LA, Pigantaro S, Volpe D. An integrative review of pediatric early warning system scores. Pediatr Nurs 2015;41(4):165–74.
13. Monaghan A. Detecting and managing deterioration in children. Paediatr Nurs 2005;17(1):32–5. DOI: 10.7748/paed2005.02.17.1.32.c964.
14. Watson A, Skipper C, Steury R, Walsh H, Levin A. Inpatient nursing care and early warning scores: a workflow mismatch. J Nurs Care Qual 2014;29(3):215–22.
15. Hogan J. Why don’t nurses monitor the respiratory rates of patients? Br J Nurs 2006;15(9):489–92.
16. Modell for kvalitetsforbedring. Oslo: Kunnskapssenteret; 2015. Available at: http://www.helsebiblioteket.no/kvalitetsforbedring/slik-kommer-du-i-gang/modell-for-kvalitetsforbedring(downloaded 05.01.2016).
17. Polit DF, Beck CT. Nursing research. 9. ed. Philadelphia, PA: Wolters Kluwer Health; 2012.
18. Johannessen A, Tufte PA, Christoffersen L. Introduksjon til samfunnsvitenskapelig metode. 4. ed. Oslo: Abstrakt forlag; 2010.
19. Lov 28. april 2017 nr. 23 om organisering av forskningsetisk arbeid (forskningsetikkloven). Available at: https://lovdata.no/dokument/NL/lov/2017-04-28-23(downloaded: 10.12.2017).
20. Hazinski MF. Children are different. In: Hazinski MF., eds. Nursing care of the critically ill child. St. Louis, MO: Mosby; 2013. s. 1–18.
21. Markestad T. Klinisk pediatri. 3. ed. Bergen: Fagbokforlaget; 2009.
22. Oliver A, Powell C, Edwards D, Mason B. Observations and monitoring: Routine practices on the ward. Paediatric Nursing 2010;22(4),28–32.
23. Cornell P, Gervis MT, Yates L, Vardaman GL. Impact of SBAR on nurse shift reports and staff rounding. Medsurg Nursing 2014;23(5):334–2.
24. Horgan M. Communication is key. WIN 2013;21(1):46–7.
25. Pearson GA, ed. Why children die: A pilot study 2006; CEMACH; 2008. Available at: http://www.publichealth.hscni.net/sites/default/files/Why%20Children%20Die%20-%20a%20pilot%20study%202006.pdf(downloaded 20.03.2016).
26. The Cosmin Checklist. VU University Medical Center. Available at: http://www.cosmin.nl/cosmin_checklist.html(downloaded 16.11.2015).
27. Bjørndal A, Hofoss D. Statistikk for helse- og sosialfagene. 2. ed. Oslo: Gyldendal Akademisk; 2004.










Comments