Registered nurses’ experiences of using ESAS to map cancer patients’ symptoms
When ESAS is routinely used to map symptoms, the patients experience greater symptom relief and a better quality of life. However, not everyone uses the tool systematically.
Background: Cancer patients in the palliative care stage often have a complex symptom profile, with many troublesome symptoms that reduce the patient’s quality of life. The patient’s experience of symptoms is subjective. A prerequisite for effective symptom relief is the systematic mapping of symptoms. Using a standardised screening tool such as the Edmonton Symptom Assessment System (ESAS) is recommended. Despite this recommendation, using ESAS in clinical practice presents a challenge. Registered nurses (RNs) play a key role in mapping and relieving symptoms and are therefore users of ESAS.
Objective: The study seeks to explore RNs’ experiences of using ESAS to map symptoms of hospital patients with cancer in the palliative care stage.
Method: We conducted qualitative in-depth interviews of six RNs from two oncology wards in a hospital in the South Eastern Norway Regional Health Authority. A semi-structured interview guide was used for the interviews. We analysed the dataset using Malterud’s systematic text condensation.
Results: The RNs consider ESAS to be a useful screening tool that provides a picture of the patient’s subjective experience of symptoms, as well as an aid for initiating conversations with patients about their symptoms, which in turn contributes to better symptom relief and quality of life for patients. Routine use is considered important for effective use of the tool. Nevertheless, ESAS is not used consistently, which impacts on the extent to which it helps to systematise the mapping of symptoms. The RNs identified several barriers to and improvement measures for routine use of ESAS.
Conclusion: If ESAS is to help systematise the mapping of symptoms, it should be used routinely. Future research should therefore focus on identifying barriers and evaluating measures that can remove the barriers that lead to the varying use of ESAS.
Patients with incurable cancer often have complex and troublesome symptoms that impact on their quality of life (1–5). Research shows that, on average, this patient group reports 9-18 symptoms (2, 3, 6). Tiredness, pain, nausea, loss of appetite, shortness of breath, anxiety, sleep problems, depression and elimination disorders are the most commonly reported (2, 6, 7).
The World Health Organization (WHO) defines palliative care as an approach designed to improve the quality of life of patients and their families in the encounter with a life-threatening illness.
Quality of life can be improved by preventing and alleviating suffering through early identification, and thorough the assessment and treatment of pain and other physical, psychosocial and spiritual problems (8). The palliative care stage is the period from when the disease is deemed incurable up to the patient’s death (1).
Systematic symptom mapping
A prerequisite for effective symptom relief is systematic symptom mapping, where the use of standardised screening tools is recommended (1, 2, 4, 9). Systematic mapping is also important because research shows that healthcare personnel tend to underestimate patients’ symptoms, resulting in undiagnosed symptoms and lack of symptom relief (2, 10, 11).
Since the patient’s experience of symptoms is always subjective, self-reporting tools are recommended as a method for enabling patients to describe their symptom experience (1, 2, 4). The national action plan for palliative cancer care (1) recommends the Edmonton Symptom Assessment System (ESAS) as the standardised screening tool.
ESAS was developed by Bruera et al. (12) in 1991 as a clinical screening tool, with the aim of improving the mapping of patients’ symptoms, identifying more symptoms and providing better quality in the treatment and care of patients (7).
ESAS is a validated and internationally recognised self-reporting tool that covers nine symptoms (pain, tiredness, drowsiness, nausea, appetite, shortness of breath, depression, anxiety and well-being) and an additional question (other problem). Symptoms are scored on a numerical scale (RNRS) from 0 to 10 (from no symptoms to worst possible severity). When mapping pain, a body diagram is used for the patient to describe the nature and location of the pain (7, 9, 12, 13).
Not everyone uses ESAS
Although it is recognised that systematic symptom mapping is essential to alleviating patients’ symptoms, getting healthcare personnel to use ESAS is a challenge (2, 4, 14–16).
According to ESAS’s clinical procedure (9), healthcare personnel who have the daily responsibility for patient treatment and care are users of ESAS. The basic task of the RN is to attend to the patient’s basic needs, and he/she is in direct contact with the patient throughout the day (5).
Oncology nursing is a common specialisation among RNs working with the palliative patient group. The palliative function of the oncology nurse includes performing nursing interventions that limit the extent of the patient’s physical, mental, social and spiritual or existential stresses.
In order to care for the patient, the RN must continuously map the patient’s resources and needs and implement and evaluate measures. Systematic symptom mapping with ESAS is therefore a key nursing task (5, 15, 17).
Earlier research
We conducted literature searches in Cinahl, Medline, Cochrane, PubMed and McMasterPluS in April–May 2017 and April–May 2018, which indicates that studies on RNs’ experiences of systematic mapping using ESAS are limited. Fitch et al. (15) support this finding.
Given the RN’s key role in mapping patients’ symptoms, there is a need to take an in-depth look at how RNs use ESAS in clinical practice.
Objective of the study
The objective of the study was to explore RNs’ experiences of using ESAS to map symptoms in hospital patients with palliative stage cancer. We wanted to answer the following research question: What experiences do RNs have of using ESAS to map symptoms in patients with palliative stage cancer?
Method
Design
The study has an interpretive qualitative design with in-depth interviews.
Sample
A strategic sample was selected. We recruited RNs from two oncology wards in a hospital in South Eastern Norway Regional Health Authority. The inclusion criteria were that the participants had to be RNs, preferably with relevant further education.
We sought to recruit RNs who work with patients with palliative stage cancer in a hospital setting. In addition, they should have knowledge and experience of symptom mapping and ESAS, at least two years of relevant work experience and be working in at least a 50 per cent FTE position.
The participants were recruited through the ward nurses, who forwarded the participant request to their RNs. The RNs’ participation was voluntary. The first author interviewed six RNs from November to December 2017.
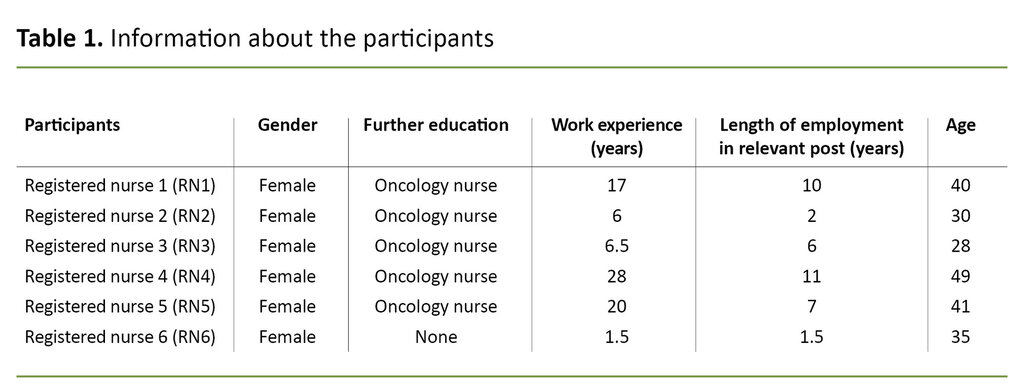
All participants were women, five of whom were oncology nurses. The average age was 37.2 years, the average work experience was 13.2 years, and the average length of employment was 6.3 years. See Table 1 for participant information.
Data collection
We developed a semi-structured interview guide (18, 19). The questions included the use of ESAS in practice, the tool’s contribution to systematic mapping, the consistency between practice and the clinical procedure, the benefits and challenges of the tool and the significance for the patient’s treatment. The interviews were conducted at the participants’ workplace and lasted 30–45 minutes. The interviews were documented in audio recordings, which were transcribed by the first author.
Analysis
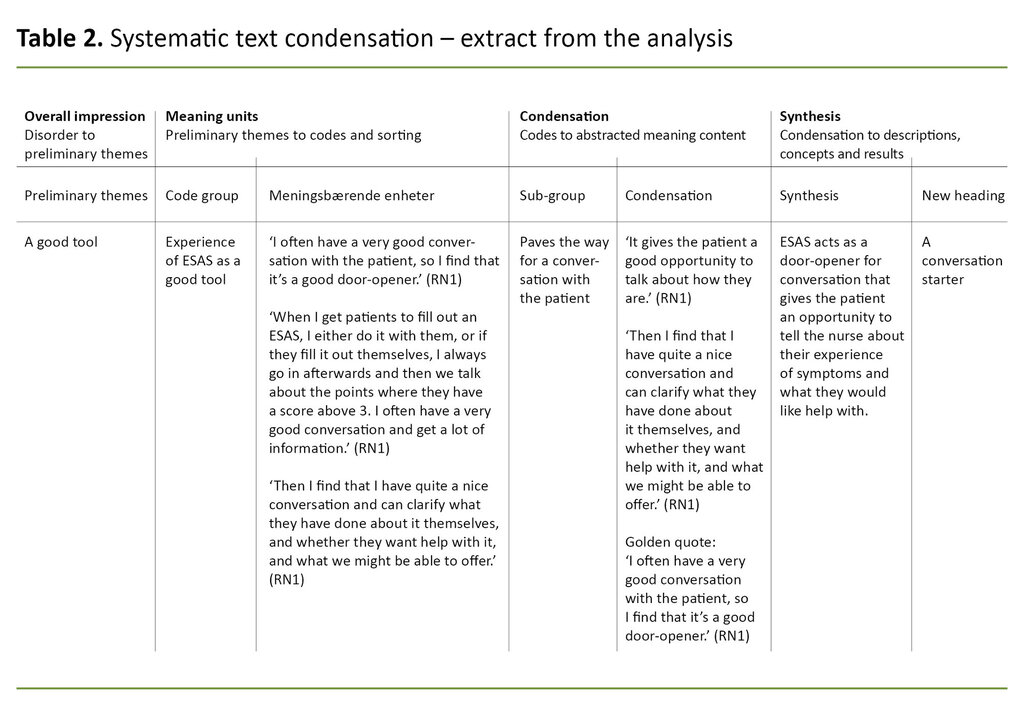
We used an interpretive cross-sectional analysis based on Malterud’s systematic text condensation. The method of analysis consists of four steps. See Table 2 for extracts from the analysis. In step 1, ‘Overall impression’, the material was read through several times in order to gain an overall impression and identify preliminary themes.
In step 2, ‘Meaning units’, we separated relevant and irrelevant text in order to identify meaning units that elucidate the research question, and sorted them into code groups.
In step 3, ‘Condensation’, we systematically extracted meaning by condensing and abstracting the content of the meaning units in the code groups and sorting these into additional sub-groups. Each sub-group was condensed, with a golden quote summarising the main elements. In the final step, ‘Synthesis’, knowledge was summarised in the form of interpretive syntheses, which formed the basis for descriptions, concepts and results (19).
Ethical considerations
The study was approved by the Norwegian Centre for Research Data (NSD), with reference number 55706. Participation was voluntary, and signed consent was obtained. All information and data have been treated confidentially, anonymised and stored in accordance with research ethics regulations (19).
Results
There were many similarities and few differences between the RNs’ experiences. Throughout the analysis phase, two overarching themes emerged with sub-points, which are: ‘a useful tool for mapping patients’ symptoms’ and ‘varying use of ESAS in clinical practice’.
A useful tool for mapping patients’ symptoms
The patient’s subjective symptom profile
ESAS is described as a useful tool for mapping patients’ symptoms: ‘ESAS contains the most important points for mapping the patient.’ (RN4) and ‘You get a good idea of how the patient is doing and what things we need to address.’ (RN3)
ESAS provides a snapshot of patients’ symptoms, and RNs can monitor these over time. The RNs highlighted in particular that ESAS throws light on the patient’s experience of their symptoms: ‘You get the patient’s subjective experience of the various symptoms.’ (RN6) and ‘The patients report their symptoms themselves. We don’t assume anything.’ (RN2).
A conversation starter
The RNs find that ESAS paves the way for a conversation in which the patient can express their symptoms, and the RN gains an overview of the patient’s symptoms and needs: ‘Not many patients tell you directly how they feel, but with ESAS it can be easier to start the conversation.’ (RN6) and ‘ESAS enables the patient to think about how they feel and have a good conversation with the nurses.’ (RN3)
The RNs also find that they obtain more information with ESAS: ‘I feel you often get to know more if you just ask ‘Hi, how are you today?’ (RN3). The RNs believe this in turn has a positive impact on the patient’s attitude towards ESAS: ‘The patient sees that what they are expressing is being taken seriously and that measures are implemented that help them. This makes them more motivated to use ESAS again.’ (RN6).
Varying use of ESAS in clinical practice
Inadequate follow-up of routines
The RNs stated that they are responsible for handing out the ESAS form, helping patients to complete it, reviewing the answers with the patients, bringing ESAS to the doctor’s visit or interdisciplinary meetings and electronically recording ESAS in the patients’ records. ESAS was most frequently used for admissions, complicated symptom profiles and in the contact with a palliative care team. The RNs emphasised the importance of routine use of ESAS and following up the symptoms reported.
Despite what the RNs told us, ESAS was used to varying degrees, and it was often just ‘another paper in the pile’. ESAS was often used when the individual RN considered it necessary: ‘We think ESAS is a good tool. We have a fixed routine for using it. We always use it at admission and one day a week. Then it should be used when there are changes. That’s the plan anyway, but I don’t think we’re always very good at following it.’ (RN1).
Lack of time was a barrier highlighted by the RNs: ‘It’s busy, and you prioritise as best you can, so ESAS is often forgotten or given a low priority.’ (RN1). In addition, it was difficult to follow up ESAS as they found it time-consuming to document the form electronically and because the body diagram cannot be documented systematically in an electronic format.
Unclear routines lead to varying practices
The routines were perceived as unclear, and the RNs expressed concern about the consequences of this: ‘Our practices vary a bit, and we often get different answers from the same patient. Since there are no clear procedures for ESAS in the ward, we may be overlooking a lot of the symptoms.’ (RN6) and ‘If the patient has never had any kind of follow-up from ESAS, they may not see the benefit of the tool, in which case they just see it as a burden.’ (RN6).
The inconsistent use of ESAS means that the RNs do not consider it to make any worthwhile contribution to systematising the symptom mapping. They also pointed out that healthcare personnel often put the emphasis on their own clinical assessments of the patient’s symptom profile, even though they often found this to be less effective than using ESAS: ‘We probably think we’re better at reading and understanding patients than we really are. When you fill out an ESAS, the patient’s symptom profile is almost always different to what we thought it was.’ (RN1).
Insufficient emphasis on using ESAS
One of the main barriers highlighted by the RNs was the lack of focus on ESAS. This barrier applied to several occupation groups, particularly doctors, who showed little interest in ESAS unless the RNs encouraged them. There were also no consequences if ESAS was not used: ‘If we don’t use ESAS, there are no repercussions for us.’ (RN5)
There was also little emphasis on ESAS in their training, and little knowledge of the clinical procedure for ESAS. The RNs’ suggestions on how ESAS can be used more routinely entailed all healthcare personnel having a greater focus on ESAS. ESAS should be a topic in the training context, and resource persons and management should show greater commitment:
‘I think we need to put more focus on training, what to use ESAS for, and how to follow up the symptoms reported.’ (RN2) and ‘If the clinical nurse educator or managers had a stronger focus on ESAS, we might automatically become better at using it.’ (RN5).
Familiarising interdisciplinary groups with ESAS was also suggested: ‘We should definitely introduce it to them more often, and the other disciplines should also perhaps have a greater focus on it.’ (RN2).
Discussion
Benefits of using ESAS
Since the patient’s experience of symptoms is always subjective, using a self-reporting tool is important for bringing the patient’s experience of symptoms to light (1). The RNs in this study find that ESAS is a useful screening tool because the form provides a snapshot of the patient’s experience of symptoms, it brings more symptoms to light, and symptoms and the efficacy of interventions can be monitored over time.
The same factors are supported in earlier literature (1, 2, 9, 20-23). Patients with a high ESAS score often have a high symptom burden (24). Patients with palliative stage cancer often experience many symptoms that can change rapidly, or over time, and can adversely affect one another (2–4). The aforementioned benefits of ESAS could therefore help to make the symptom mapping more systematic and effective.
At the same time, misunderstandings about ESAS can impact on how it is used, and where this is the case, the form may not represent the patient’s real experience of symptoms. One such example is the misinterpretation of terms such as ‘drowsiness’ and ‘tiredness’. Many find it difficult to understand the difference between these.
Another example is the misinterpretation of the NRS scale. In ESAS, ‘appetite’ is ranked from 0 = best appetite to 10 = worst possible appetite. Patients tend to believe that a high score means a good appetite (2). Appropriate use therefore requires RNs to have adequate knowledge of ESAS and to be able to guide the patient.
Good communication important
Ascertaining the patient’s experience of symptoms depends on good communication between the patient and the RNs (1). The RNs in this study find that ESAS paves the way for a conversation with the patient about their symptoms. Their experiences are consistent with findings from earlier research (1, 9, 20–22, 25).
Since ESAS does not cover all the symptoms the patient may experience, ESAS should be used as a starting point for conversation (20, 21). By starting the conversation about ESAS, the RN can identify more symptoms, including those not covered in the form. They can then go into more depth about the nature of the symptoms and how they affect the patient, which in turn can help the patient to feel that they are being acknowledged and taken seriously.
If the RNs do not use a systematic mapping tool, the patient’s symptoms may be overlooked. ESAS is a tool and a starting point. In order to make a comprehensive assessment of the patient’s symptom profile, the RNs also need to use their professional judgement (26).
ESAS must be used routinely
The RNs in this study are concerned that ESAS must be used routinely if the mapping of symptoms is to be systematic. This view is supported by national guidelines and the clinical procedure (1, 9). Research shows that many symptoms are not reported by patients and are therefore not identified. Using self-reporting tools such as ESAS will therefore play a vital role in mapping symptoms (6).
The results can therefore be interpreted to mean that routine use of the form is a prerequisite for enabling the systematic mapping of symptoms, and in turn better symptom relief. This correlation is supported by Chen and Hollis (27), who recommend the routine use of self-reporting tools such as ESAS as they foster better communication between the patient and the healthcare personnel.
Furthermore, the systematic mapping of symptoms means that more symptoms are identified, the efficacy of interventions can be evaluated, and patient satisfaction with interventions increases.
Counteracting barriers to using ESAS
The RNs in this study and other studies emphasise that mapping and providing relief for the patient’s symptoms is one of their areas of responsibility (20-22). In this study, the RNs explain how important it is to use ESAS routinely, and the responsibility they have for the effective mapping of symptoms.
Why the RNs’ use of the form varies to the extent that it does when they are so aware of the benefits of ESAS is unclear. The RNs’ experiences differ here from other studies, where the majority report that they always or almost always use ESAS and encourage patients to use it (20–22).
The RNs note that their varying use may mean that some of the patients’ symptoms do not come to light. It is also indicated that their own clinical assessments are often given priority over assessments made in ESAS. Research shows that healthcare personnel often underestimate the patient’s symptoms, which strengthens the argument for using a self-reporting tool (2, 10, 11).
When the RN’s mapping of symptoms does not reflect the patient’s actual symptoms, it is difficult to implement targeted symptom relief interventions. Furthermore, it will be difficult to monitor the patient’s symptoms over time if ESAS is not used routinely.
It can therefore be said that varied use reduces the usefulness and usability of the form. It is therefore important that the RNs understand the responsibility they have when using ESAS, and that they realise that inconsistent use can counteract the intention of the form.
The RNs point out several potential barriers and measures for routine use of ESAS. These are consistent with findings in earlier research (2, 10, 14–16, 20–23, 25, 28, 29). The results may indicate that there is a need to identify and address barriers that prevent routine use if ESAS is to contribute to the systematic mapping of symptoms.
RNs’ experiences are supported by several systematic literature reviews (14, 16). The reviews emphasise the importance of identifying barriers that hamper and factors that promote the implementation and use of self-reporting tools such as ESAS in palliative practice.
Although RNs recognise the value of ESAS, their hectic working days make it difficult to use the tool routinely, particularly when procedures for using ESAS are perceived as unclear and failure to use it has no repercussions for healthcare personnel. Routine use of ESAS becomes particularly difficult when it is up to each nurse to evaluate how ESAS should be used and whether they should use ESAS consistently.
Additionally, adhering to the procedures can be difficult when RNs cannot rely on nurses on the next shift to follow up on ESAS. The RNs’ apparent responsibility for engaging other occupational groups, and managers’ and/or resource persons’ lack of interest in ESAS both represent challenges.
Conversely, if the RNs knew that managers, resource persons, other occupation groups or nursing colleagues expected ESAS to form part of the reporting or patient record-keeping, ESAS would become a natural tool that is easy and necessary to use. The results may therefore indicate that future research should focus on identifying barriers and evaluating preventive measures if ESAS is to contribute to the systematic mapping of symptoms.
Limitations of the study
The results from a study with six informants cannot be generalised. The informants are from oncology wards, but we may have had a somewhat varying result if the informants were from purely palliative care wards. Another factor is that the majority of the informants had extensive work experience and a relevant specialisation. It is conceivable that a study with RNs who have less experience and/or no specialisation could have a different result.
Nevertheless, we must be able to assume that the findings are transferrable to similar settings to some degree and can therefore have external validity (18). Since informants use the tool in their daily work, we must also be able to assume that their experiences are credible and therefore have an intrinsic validity (19). Furthermore, the informants have knowledge of and experience in using ESAS, which should contribute to the reliability of the study (19).
Finally, it must be mentioned that we work in the field of palliative care. This may have been an obstacle in interpreting the RNs’ responses, but may also have been a positive opportunity to understand their perspectives (18, 19).
Conclusion
ESAS is described as a useful tool for mapping symptoms in the relevant patient group, which in turn promotes better symptom relief and quality of life for patients. Routine use is emphasised for fulfilling the intention of the form. Nevertheless, ESAS is used to varying degrees, which affects the extent to which the tool contributes to the systematic mapping of symptoms.
The study points to several barriers to and measures for routine use of ESAS. Qualitative research on the topic is limited, and further research is needed in the field. The results of this study suggest four factors that may improve the use of ESAS in practice:
- Clarify ESAS procedures and obtain a consensus on their use throughout the ward.
- Increase the focus on ESAS in training to ensure a shared understanding across the different disciplines of the purpose, relevance and use of the tool.
- Raise awareness of the individual’s and every occupation group’s responsibility for ESAS based on their expected area of responsibility.
- Obtain a clear commitment from resource persons and managers to promote the routine use of ESAS, where consequences for non-use are visible.
References
1. Helsedirektoratet. Nasjonalt handlingsprogram for palliasjon i kreftomsorgen. Nasjonal faglig retningslinje. Oslo; 2015. IS-2800. Available at: https://helsedirektoratet.no/retningslinjer/nasjonalt-handlingsprogram-med-retningslinjer-for-palliasjon-i-kreftomsorgen (downloaded 10.05 2018).
2. Chang VT, Smith TJ, Savarese DM. Approach to symptom assessment in palliative care. Alphen aan den Rijn: Wolters Kluwer; 2018. Available at: https://www.uptodate.com/contents/approach-to-symptom-assessment-in-palliative-care (downloaded 20.05 2018).
3. Deshields TL, Potter P, Olsen S, Liu J, Dye L. Documenting the symptom experience of cancer patients. Journal of Supportive Oncology. 2011;9(6):216–23.
4. Kaasa S, Loge JH. Diagnostikk innen palliasjon – en generell innføring. In: Kaasa S, Loge JH, eds. Palliasjon: Nordisk lærebok. 3. ed. Oslo: Gyldendal Akademisk; 2016. p. 202–16.
5. Sæteren B. Omsorg for alvorlig syke og døende pasienter: sorg og sorgarbeid. Kreftsykepleie: pasient, utfordring, handling. 4. ed. Oslo: Cappelen Damm Akademisk; 2017. p. 224–50.
6. White C, McMullan D, Doyle J. «Now that you mention it, doctor … »: symptom reporting and the need for systematic questioning in a specialist palliative care unit. Journal of Palliative Medicine. 2009;12(5):447.
7. Hui D, Bruera E. The Edmonton Symptom Assessment System 25 years later: past, present, and future developments. Journal of Pain and Symptom Management. 2017;53(3):630–43.
8. World's Health Organization (WHO). WHO definition of palliative care. Geneva; 2002. Available at: http://www.who.int/cancer/palliative/definition/en/ (downloaded 20.05 2018).
9. Oslo universitetssykehus HF. ESAS – Edmonton Symptom Assessment System. Oslo; 2018. Available at: http://www.helsebiblioteket.no/fagprosedyrer/ferdige/esas-edmonton-symptom-assessment-system--260510 (downloaded 01.07.2018).
10. Rhondali W, Hui D, Kim SH, Kilgore K, Kang JH, Nguyen L, et al. Association between patient-reported symptoms and nurses' clinical impressions in cancer patients admitted to an acute palliative care unit. Journal of Palliative Medicine. 2012;15(3):301.
11. Laugsand EA, Sprangers MAG, Bjordal K, Skorpen F, Kaasa S, Klepstad P. Health care providers underestimate symptom intensities of cancer patients: a multicenter European study. Health and Quality of Life Outcomes. 2010;8:104.
12. Bruera E, Kuehn N, Miller MJ, Selmser P, Macmillan K. The Edmonton Symptom Assessment System (ESAS): a simple method for the assessment of palliative care patients. Journal of Palliative Care. 1991;7(2):6–9.
13. Watanabe SM, Nekolaichuk CL, Beaumont C. The Edmonton Symptom Assessment System, a proposed tool for distress screening in cancer patients: development and refinement. Psycho‐Oncology. 2012;21(9):977–85.
14. Antunes B, Harding R, Higginson IJ. Implementing patient-reported outcome measures in palliative care clinical practice: a systematic review of facilitators and barriers. Palliative Medicine. 2014;28(2):158–75.
15. Fitch MI, Howell D, McLeod D, Green E. Screening for distress: responding is a critical function for oncology nurses. Canadian Oncology Nursing Journal. 2012;22(1):12.
16. Sommerbakk R, Haugen DF, Tjora A, Kaasa S, Hjermstad MJ. Barriers to and facilitators for implementing quality improvements in palliative care – results from a qualitative interview study in Norway. BMC Palliative Care. 2016;15(1).
17. Reitan AM. Kreftsykepleie. In: Reitan AM, Schølberg T, eds. Kreftsykepleie: pasient – utfordring – handling. 4. ed. Oslo: Cappelen Damm Akademisk; 2017. p. 33–47.
18. Malterud K. Kvalitative metoder i medisinsk forskning: en innføring. 4. ed. Oslo: Universitetsforlaget; 2017.
19. Christoffersen L, Johannessen A, Tufte PA, Utne I. Forskningsmetode for sykepleierutdanningene. Oslo: Abstrakt forlag; 2015.
20. Green E, Yuen D, Chasen M, Amernic H, Shabestari O, Brundage M, et al. Oncology nurses' attitudes toward the Edmonton Symptom Assessment System: results from a large cancer care Ontario study. Oncology Nursing Forum. 2017;44(1):116–25.
21. Pereira JL, Chasen MR, Molloy S, Amernic H, Brundage MD, Green E, et al. Cancer care professionals' attitudes toward systematic standardized symptom assessment and the Edmonton Symptom Assessment System after large-scale population-based implementation in Ontario, Canada. Journal of Pain and Symptom Management. 2016;51(4):662–72.e8.
22. Bainbridge D, Seow H, Sussman J, Pond G, Martelli-Reid L, Herbert C, et al. Multidisciplinary health care professionals' perceptions of the use and utility of a symptom assessment system for oncology patients. Journal of Oncology Practice. 2011;7(1):19.
23. Myhra CB, Grov EK. Sykepleieres bruk av Edmonton Symptom Assessment Scale (ESAS): palliativ behandling. Sykepleien Forskning. 2010;5(3):210–8. DOI: 10.4220/sykepleienf.2010.0113
24. Selby D, Chakraborty A, Myers J, Saskin R, Mazzotta P, Gill A. High scores on the Edmonton Symptom Assessment Scale identify patients with self-defined high symptom burden. Journal of Palliative Medicine. 2011;14(12):1309–16.
25. Carli Buttenschoen D, Stephan J, Watanabe S, Nekolaichuk C. Health care providers' use and knowledge of the Edmonton Symptom Assessment System (ESAS): is there a need to improve information and training? Supportive Care in Cancer. 2014;22(1):201–8.
26. Martinsen K. Samtalen, skjønnet og evidensen. Oslo: Akribe; 2005.
27. Chen J, Ou L, Hollis SJ. A systematic review of the impact of routine collection of patient reported outcome measures on patients, providers and health organisations in an oncologic setting. BMC Health Services Research. 2013;13(1).
28. Dudgeon D, King S, Howell D, Green E, Gilbert J, Hughes E, et al. Cancer Care Ontario's experience with implementation of routine physical and psychological symptom distress screening. Psychooncology. 2012;21(4):357.
29. Beddard-Huber E, Jayaraman J, White L, Yeomans W. Evaluation of the utility of the Edmonton Symptom Assessment System (revised) Scale on a tertiary palliative care unit. Journal of Palliative Care. 2015;31(1):44–50.










Comments