Screening for symptoms of depression associated with heart disease
Heart disease increases the risk of depression. How can we best identify depressed cardiac patients?
Background: Depression is associated with an increased risk of death from heart disease and has major implications for quality of life, working capacity and ability to comply with medical treatment. Therefore, health authorities recommend routine screening for depression.
Purpose: The purpose of the study was to test a method of mapping symptoms of depression in patients with heart disease.
Method: We tested a method of screening for depression in 106 patients with cardiovascular disease in a cardiac medical ward, using the Patient Health Questionnaire (PHQ-9). The most common diagnoses on admission were arrhythmia, heart failure and valvular heart disease.
Result: Compared with previous studies, we found far fewer patients who met the criteria for depression and concomitant heart disease. Only six out of 106 satisfied these criteria, while a significant subset of 33 patients had mild symptoms of depression.
Conclusion: Patients with heart disease are at risk of developing depression, and the patients in question must be identified so that they can receive appropriate health assistance. The study showed that it was possible to implement a method of screening for symptoms of depression in a cardiac medical ward using PHQ-9. It may be the case that screening at an acute phase of heart disease is premature. We are now conducting a study of screening procedures during the outpatient follow-up after discharge.
The study described in this article is based on three key facts:
- Heart disease gives increased risk of depression.
- Depression has serious consequences for the sick person.
- The health service is not sufficiently proficient at identifying and implementing measures to combat depression in connection with heart disease.
Background
The prevalence of depression associated with heart disease is estimated at between 15 and 40 per cent (1). There is considerable variation linked to the type of study and whether self-report questionnaires or structured diagnostic interviews are used. Nonetheless, depression is consistently found to occur far more often among those who are medically ill than in the healthy population, with a two to three times higher estimated prevalence (2). Studies at somatic hospitals indicate that 15 to 20 per cent of patients with myocardial infarction have a depressive disorder consistent with diagnostic criteria, but an even greater number have an increased level of symptoms of depression. The same prevalence is reported among patients hospitalised with other kinds of heart disease (3). Outpatients also exhibit a clearly higher prevalence than people who do not have cardiovascular disease. An American study of 30 000 adults found a prevalence of 9.3 per cent among patients with cardiovascular disease over a 12-month period, and 4.8 per cent for those with no concomitant medical disorders (4).
Major negative consequences
Depression can have a profound effect on the course and outcome of heart disease and is associated with non-compliance with treatment and rehabilitation programmes (3). When reviewing approximately 150 000 participants in 54 studies, Nicholson et al. found that depression predicted a cardiovascular event in people who had not previously had heart disease and new events for those who had. Moreover, the risk of major negative consequences was twice as great following a cardiovascular event (5).
The report by Folkehelseinstituttet [Norwegian Institute of Public Health] on mental disorders in Norway from a public health perspective (6) states that depression is as strong a risk factor for death as smoking. This applies in particular to mortality resulting from cardiovascular disease and infectious diseases. The report refers to three correlations between depression and death resulting from cardiovascular disease. Firstly, depressed patients smoke more than the rest of the population. They drink more alcohol, train less and have a poorer diet. Secondly, depression may be a result of the burden of somatic illness. However, the risk of serious illness also increases with depression. Thus, the negative impact has a two-way effect. Thirdly, a negative interaction may arise that in itself leads to increased mortality due to depression and heart disease since depression weakens the body’s powers of resistance.
Greatest impact on working capacity
Patients with cardiovascular disease and concomitant depression demonstrate three times less compliance with recommended medical treatment than patients without depression (7). This entails reduced health and life quality, increased financial expenses and at worst a shorter life expectancy. For the health service, this means more consultations, longer periods of hospitalisation and more readmissions.
Depression is a major cause of increased sickness absence in the case of somatic illness (8) and can be the determinant that excludes the person concerned from the labour market (9). An American study of 20 000 employees who were treated for depression showed that depression affected working capacity more negatively than all other illnesses (10). Depression may result in withdrawal from social life and increased irritability and pessimism, which in turn affects relationships with managers and colleagues. Some employees may become less productive due to a decline in energy, poorer concentration and a reduced ability to make decisions (11).
Depression is underdiagnosed and undertreated in patients with concomitant somatic illness. In line with the American Heart Association (3) and a number of European guidelines (12), Helsedirektoratet [Norwegian Directorate of Health] (13) recommends screening routines for depression. How can we make provision for this in a busy hospital everyday setting, and at what point during the treatment should we conduct such screening? The purpose of our study is to test and evaluate a method for systematic screening for depression in patients in cardiac medical wards. Early identification may enable mental health assistance to be provided at an earlier stage.
Method
We conducted the study in cooperation with the cardiac medical ward (professional nurses, head of the unit and senior consultant) and specialists in psychology at the Diakonhjemmet Hospital’s Unit for Mental Health Services in Somatic Care. The specialists in psychology lectured on depression as well as training health personnel on the ward in the use of the screening tools. The specialist nurses on the ward were of key importance in developing specific procedures for when, how and where screening should take place, and they administered the collection of data. A specialist in psychology and a specialist nurse carried out interviews with patients in the further screening of those assessed as possibly depressed (score on Beck’s depression inventory scale (BDI-II) was 14 or more). For practical reasons, we tested the screening over two six-week periods.
Inclusion and exclusion criteria
We assessed all the patients admitted to the cardiac medical ward with a heart-related diagnosis with a view to their participation in the study. We excluded patients who had been admitted with a myocardial infarction, and who were already included in another research project. The exclusion criteria were language problems, reduced reading ability and inability to complete the questionnaire. In addition, we excluded patients who were too ill, or who were unable to participate for other practical reasons. We asked a total of 154 patients, 48 of whom declined to participate in the survey. Out of 106 participants, just over half were men (58 patients). The reasons for refusal to participate were complex. Some were too tired to complete the questionnaire mapping symptoms of depression (Patient Health Questionnaire-9 (PHQ-9)), some did not wish to participate, whilst others had been discharged before we offered screening. The most common diagnoses on admission were arrhythmia, heart failure and valvular heart disease. The Regional Committee for Medical and Health Research Ethics (REC) has approved the project.
Screening instrument
We asked the patients taking part to fill in PHQ-9 – a questionnaire with nine diagnostic criteria for depression (14) (table 1). Like other well-documented questionnaires, PHQ-9 has fairly high accuracy with a sensitivity of up to 80–90 per cent (15), while some studies have exhibited lower sensitivity (16). It takes 2–5 minutes to complete the form. Each question has four possible responses from 0 to 3 (0 = Not at all, 1 = Several days, 2 = More than half the days and 3 = Nearly every day). A score of 10 or more indicates possible depression and a need for further screening (16).
Further screening
A nurse carried out further screening of the patients with a score of 10 or more on the PHQ-9 questionnaire. The screening was based on Beck’s depression inventory scale (BDI-II), one of the most commonly used self-report scales for level of depression, i.e. degree of symptoms of depression (17). In addition, we asked patients about earlier depression, grief reactions and social support. Patients with symptoms of depression were given the opportunity to meet with a psychologist at the hospital, or were referred to the outpatient clinic for follow-up. We conducted a cluster analysis to explore possible patterns and profiles related to PHQ-9 scores in accordance with the k-means clustering method, whereby the values of a sample of variables are analysed to identify any sub-groups (18).
Results
Of the 106 patients who were screened for depression, 19 (18 per cent) had a PHQ-9 score of 10 or more, and underwent further screening for depression. Seven of these (7 per cent of the total number of screened patients and 37 per cent of the patients with a PHQ-9 score of 10 or more) had a BDI-II score of 14 or more, and had a feedback session with a psychologist. Six of these patients (6 per cent of the total number of screened patients and 32 per cent of the patients with a PHQ-9 score of 10 or more) were diagnosed with depression. The cluster analysis showed that a group of 33 participants had a relatively high mean score on questions about sleeping problems and tiredness.
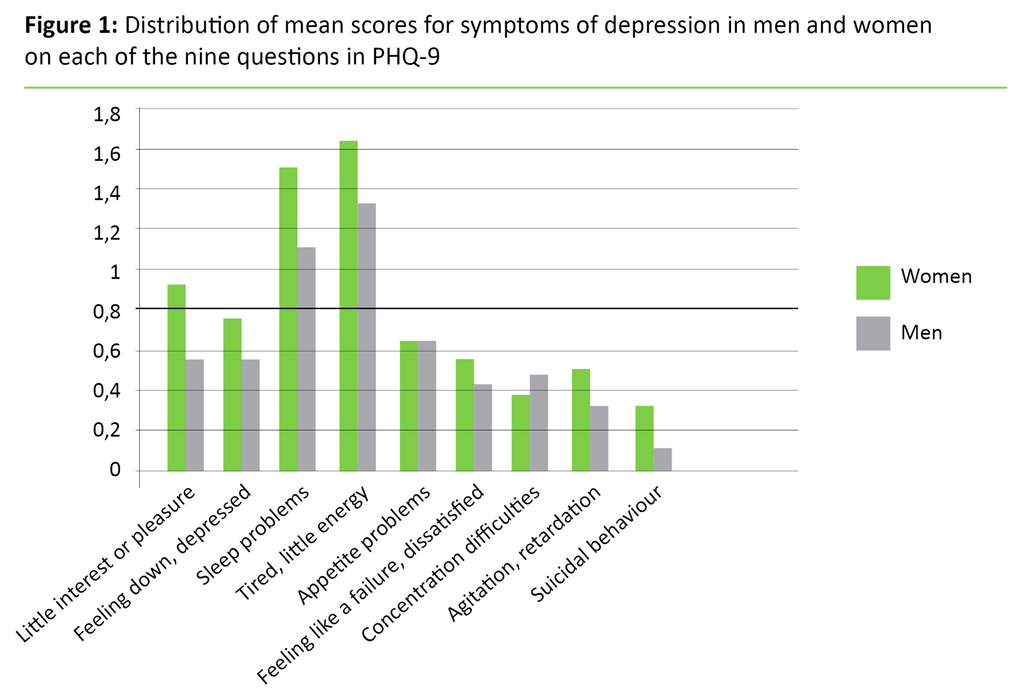
Figure 1 shows the distribution of mean scores for women and men for each of the nine questions. All nine questions for both sexes gave 0 as the minimum value and 3 as the maximum value. Women tended to score higher than men on most of the questions but the disparities were not statistically significant for any of the questions. Nor were there any statistically significant gender disparities for the total score on PHQ-9.
Discussion
We were able to implement the screening method we tested in the study in a systematic manner. However, a large number of responses were lacking (48 patients), which may have been due to the fact that it is challenging to carry out screening during the short period of admission to the ward (two days on average). It may be difficult for many patients to answer the questionnaire and questions on their mental health during this acute medical phase. Moreover, since this is a research project, patients had to deal with a written consent form in addition to verbal information about the study.
An alternative to using the questionnaire may be to ask the first two questions in PHQ-9, referred to as PHQ-2, orally, as recommended by Helsedirektoratet [Norwegian Directorate of Health] (13): ‘Over the past two weeks, have you been feeling down, depressed or hopeless?’ and ‘Over the past two weeks, have you often felt little interest or pleasure in doing things?’ If the patient responds affirmatively to these questions, further questions about symptoms of depression can be posed. This method can work just as well as using questionnaires (19). However, using PHQ-2 might have captured a larger number of those who reserved the right to refuse to participate.
Should be examined at a later stage
When we compared prevalence with previous studies, we found fewer patients who satisfied the criteria for depression and concomitant heart disease – only six out of 106. A weakness of our study is that patients with acute myocardial infarction are not included, since this group exhibits a high incidence of depression (20). Another weakness may be that the PHQ-9 screening instrument was not sufficiently sensitive, and we know of no validation studies among patients with heart disease in Norway. Another explanation for the identification of only a few patients with depression may be the time of the screening, since this was undertaken at an acute phase when the patients were tired and distressed with regard to having heart disease or aggravated symptoms, while they were also happy about having survived. The hospital’s self-management courses for patients with cardiovascular diseases and family members reveal that some people only report problems of depression after they have been at home for a period of time. It will be interesting to conduct follow-up Norwegian surveys of screening for symptoms of depression in hospitalised patients at some point after they have been discharged from hospital. We are now in the process of conducting such a study.
How did patients react to questions about depression? Patients who proved to be depressed expressed their gratitude that this had been detected and that they had been offered follow-up. Meanwhile, several did not wish to participate because they were tired. They said that they had more than enough to think about without also having to fill in a questionnaire on depression.
A number of patients reported symptoms of depression, but did not meet the criteria for clinical depression. A total of 33 patients had considerable problems with sleep and exhaustion, and in some cases other mild symptoms of depression. It is important to also pay attention to mild symptoms because they undermine life quality, reduce functional level and can diminish compliance with recommended measures (15). Moreover, patients who experience considerable tiredness and sleeping problems may be at risk of developing depression at a later stage. Prevention is vital in this respect, for example by informing the patient, family members and GP about the increased risk, and informing them of the measures described in the guidelines of the Helsedirektoratet [Norwegian Directorate of Health] (13).
Conclusion
Screening for symptoms of depression in a cardiac medical ward requires methods that are straightforward and easy to implement because the period of hospitalisation is short and the hospital personnel have a hectic working day. The study shows that it is possible to carry out screening using the PHQ-9 questionnaire in line with the recommendations of professional guidelines. A possible alternative is to ask PHQ-9’s first two questions, also referred to as PHQ-2, orally. Many patients with heart disease report signs of depression after they have been at home for a period of time following discharge, when they are trying to get their lives back on track. It is vital to put in place screening procedures later in the course of the illness.
We are in the process of initiating a new study in which we map symptoms of depression at the outpatient follow-up, using PHQ-2 and another scale for level of depression: the Hospital Anxiety and Depression Scale (HADS). The latter form replaces BDI-II, which we used in this study. HADS gives us the opportunity to establish whether an anxiety disorder is present. Heart disease combined with anxiety is associated with increased risk of aggravation of heart disease (21, 22).
Referanser
1. Kop WJ, Plumhoff JE. Depression and coronary heart disease: Diagnosis, predictive value, biobehavioral mechanisms, and intervention. In: Allan R, Fisher J (ed.). Heart and mind. The practice of cardiac psychology. Washington DC: American Psychological Association 2012:143–68.
2. Creed F, Dickens C. Depression in the medically ill. I: Steptoe S (red.). Depression and physical illness. Cambridge University Press 2007:3–18.
3. Lichtman JH, Bigger JT, Blumenthal JA et al. Recommendations for screening, referral, and treatment: A science advisory from the American Heart Association Prevention Committee of the Council on Cardiovascular Nursing, Council on Clinical Cardiology, Council on Epidemiology and Prevention, and Interdisciplinary Council on Quality of Care and Outcomes Research: Endorsed by the American Psychiatric Association. Circulation 2008;118:1768–75.
4. Egede LE. Major depression in individuals with chronic medical disorders: prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen Hosp Psych 2007;29:409–16.
5. Nicholson A, Kuper H, Hemingway H. Depression as an aetiologic prognostic factor in coronary heart disease: A meta-analysis of 6362 events among 146,538 participants in 54 observational studies. Eur Heart J 2006;27:2763–74.
6. Nasjonalt folkehelseinstitutt. Psykiske lidelser i Norge: Et folkehelseperspektiv. Rapport 8:2009. Oslo: Nasjonalt folkehelseinstitutt 2009.
7. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: Meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med 2000;160:2101–7.
8. Soderman E, Lisspers J, Sundin O. Depression as a predictor of return to work in patients with coronary artery disease. Soc Sci Med 2003;56:193–202.
9. Henderson M, Harvey SB, Øverland S, Mykletun A, Hotopf M. Work and common psychiatric disorders. J R Soc Med 2011;104:198–207.
10. Kessler R, White LA, Birnbaum H, Qiu Y, Kidolezi Y, Mallett D, Swindle R. Comparative and interactive effects of depression relative to other health problems on work performance in the workforce of a large employer. J Occup Environ Med 2008;50:809–16.
11. Berge T, Falkum E. Se mulighetene. Psykisk helse og arbeidsliv. Oslo: Gyldendal Akademisk 2013.
12. Graham I, Atar D, Borch-Johnsen K et al. European guidelines for cardiovascular disease prevention in clinical practice: executive summary. Eur Heart J 2007;28:2375–2414.
13. Nasjonale retningslinjer for diagnostisering og behandling av voksne med depresjon i primærhelsetjenesten og spesialisthelsetjenesten. Helsedirektoratet 2009.
14. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001;16:606–13.
15. U.S. Preventive Services Task Force. Screening for depression: Recommendations and rationale. Ann Intern Med 2002;136:760–64.
16. McManus D, Pipkin SS, Whooley MA. Screening for depression in patients with coronary heart disease (data from the Heart and Soul Study). Am J Cardiol 2005;96:1076–81.
17. Beck AT, Steer RA, Brown GK. Beck Depression Inventory – Second Edition. Harcourt Assessment, Inc., USA. 1996.
18. SPSS Statistics 22.0.0. K-means Cluster Analysis. Available at: http://www-01.ibm.com/support/knowledgecenter/SSLVMB_22.0.0/com.ibm.spss.statistics.help/spss/base/idh_quic.htm (downloaded 23.03.2016).
19. Henkel V, Mergl R, Coyne JC, Kohnen R, Moller HJ, Hegerl U. Screening for depression in primary care: will one or two items suffice? Eur Arch Psychiatry Clin Neurosci 2004;254:215–23.
20. Lichtman JH, Froelicher, ES, Blumenthal JA et al. Depression as a risk factor for poor prognosis among patients with acute coronary syndrome: Systematic review and recommendations. A scientific statement from the American Heart Association. Circulation 2014;129:1350–69.
21. Janszky I, Ahnve S, Lundberg I et al. Early-onset depression, anxiety and risk of subsequent coronary heart disease. 37-year follow-up of 49,321 young Swedish men. Am J Cardiol 2010;56:31–6.
22. Roest AM, Martens EJ, de Jone P et al. Anxiety and risk of incident coronary heart disease. A meta-analysis. Am J Cardiol 2010;56,38–46.











Comments