The association between depressive symptoms and self-management in persons with type 2 diabetes
It may be beneficial to screen at-risk individuals for depressive symptoms and recognise that they may need more support from healthcare personnel.
Background: Type 2 diabetes mellitus is the most common type of diabetes in the world and in Norway. People with type 2 diabetes have a significantly increased risk of developing depressive symptoms. Depression in people with diabetes is associated with adverse effects, such as increased risk of complications, poorer quality of life and, in the worst case, premature death.
Objective: To assess the occurrence of depressive symptoms in people with type 2 diabetes who were included in a mobile health intervention. We also wanted to examine the association between depressive symptoms and self-management.
Method: This is a cross-sectional study based on baseline data from a Norwegian randomised controlled trial in the EU project RENEWING HEALTH. The analyses in this study are based on baseline data from the participants in the Norwegian trial, and the participants’ data are analysed as one group (n = 151).
Results: A total of 34/149 (22.5 per cent) of participants had symptoms of depression (CES-D ≥ 16) at the time of inclusion. We found that those who reported depressive symptoms scored lower on self-management (measured using heiQ). We also found that younger people more frequently reported depressive symptoms (p = 0.042). Those reporting depressive symptoms were eight times more likely to be unemployed than those without depressive symptoms.
Conclusion: Approximately one-fifth of our sample of adults with type 2 diabetes reported depressive symptoms, and this group scored lower on self-management. National clinical guidelines for diabetes discuss whether people with type 2 diabetes and depressive symptoms should be screened for depressive symptoms in order to improve the provision of health services offered to them.
Diabetes is a chronic disease with many adverse effects for the individual’s life and health. About 90 per cent of those with diabetes have type 2 diabetes (1).
Approximately 424.9 million people are estimated to be living with diabetes worldwide, and as many as 212.4 million (50 per cent) in the age range 20–79 may be unaware that they have the condition.
The number of people with diabetes in Norway is also increasing, and around 216 000 people have a known type 2 diabetes. In 2016, around 183 000 people were taking glucose-lowering medications (2).
The association between diabetes and symptoms of depression
An association has also been shown between diabetes and symptoms of depression (3).
People with diabetes have a significantly increased risk of developing depressive symptoms compared to those without diabetes (4), and it has been shown that depression in people with diabetes may be associated with poor metabolic control (hyperglycaemia) (3), higher risk of complications (1, 3), unemployment (5), poorer quality of life (4, 5) and premature death (6).
These findings are particularly alarming when we know that people with type 2 diabetes and depression are more likely to have an incapacity for work than those with diabetes or depression alone (7).
Depressive symptoms can also reduce these people’s capacity for self-management (3). Self-management can be defined as ‘the individual’s ability to manage the symptoms, treatment, physical and psychosocial consequences and life style changes inherent in living with a chronic condition.’ (8).
The self-management of diabetes and adherence to the recommended treatment regimen is important for promoting health and preventing or delaying complications associated with diabetes (1, 9).
The individual’s capacity for self-management could be adversely affected if the therapist is unaware of the psychosocial factors associated with the illness, since people with depression may need more psychosocial support than those without depression (10).
Screening for symptoms of depression
It is therefore recommended that people with symptoms of depression are screened for psychosocial factors. One way of doing this is using questionnaires in which individuals self-report their attitudes to the disease, treatment expectations, treatment effect, quality of life and available resources (9).
In order to reduce hospital admissions and premature deaths, the International Diabetes Federation (IDF) also proposes screening people with diabetes for depressive symptoms using a validated questionnaire with some simple questions about psychological factors (11).
In Norway, there are national clinical guidelines for diabetes that steer the management of diabetes. These guidelines stipulate that there is limited evidence to support the need to screen for depression in people with diabetes, and it is therefore recommended that screening should only be conducted where there is a clinical suspicion of depression (12).
Objective of the study
The objective of this study was to assess the incidence of depressive symptoms in people with type 2 diabetes who were included in a mobile health intervention. We also wanted to examine the association between depressive symptoms and self-management.
The following research question was formulated: What is the occurrence of depressive symptoms in a given sample of people with type 2 diabetes, and what associations are there between the occurrence of depressive symptoms and participants’ sociodemographic and clinical characteristics and their self-management of the disease?
Method
Design of the study
The study is a cross-sectional study based on baseline data from the Norwegian randomised controlled trial in the EU project RENEWING HEALTH. In the Norwegian trial, we evaluated a mobile app with a diabetes diary among people with type 2 diabetes, with or without health counselling from a diabetes nurse.
We conducted a three-arm, randomised controlled trial, which has been described in detail previously (13–15). In the current study, the results were based solely on baseline data from all participants in the Norwegian trial, and we analysed the participants’ data as one group (n = 151).
Participants and recruitment
Participants were recruited in the period March 2011 to October 2012. All were living at home in the south or north of Norway and were recruited by their GP.
Some were recruited from the municipal volunteer centres, where they had been referred by their GP with a view to making lifestyle changes. Participants were also recruited from the introductory diabetes course Diabetes startkurs, which is offered by the specialist health service to newly diagnosed people living at home with diabetes in Norway (12).
The inclusion criteria were ≥18 years of age, a long-term blood glucose (HbA1c) level ≥ 7.1 per cent (54.1 mmol/mol) and being diagnosed with type 2 diabetes more than three months before the study started.
It was also important that the participants had the cognitive ability to complete the trial, and that they could use a mobile phone. Serious physical or mental illness were exclusion criteria.
Prior to the start of the project, we discussed whether some of the participants should receive a higher intensity intervention than others, including those with depression or depressive symptoms.
However, adapting the intervention based on any depressive symptoms would require more resources than we had available, so no such adaptation was made in the project.
A total of 151 participants were included, and we collected data at baseline before the participants were randomised into one of the two intervention groups or the control group.
Procedure
The doctors and others involved in the recruitment received information from us in advance about the various criteria for participation in the trial. We collected data using self-reported questionnaires.
Some clinical variables were obtained from the participants’ GPs. HbA1c was either obtained from the participants’ GP or using a HbA1c device that we had available to ensure that the procedure was as uniform as possible.
The variables used
In this cross-sectional study, we report on a selection of the variables from the randomised controlled trial, which has already been described in detail (13–15).
Sociodemographic and clinical variables
The sociodemographic data were obtained from the self-reported questionnaires developed in the EU project, and consisted of personal data such as age, gender, marital status, education and employment status.
The employment status variable was constructed as a dichotomous variable as persons in employment (including pensioners) and persons currently unemployed. Some clinical variables were self-reported, such as concurrent illnesses (comorbidity), hypoglycaemia, diabetes duration and self-monitoring of blood sugar.
The remaining clinical characteristics were obtained from the GPs, including: HbA1c, weight, height, blood pressure and medication.
Lifestyle variables
Lifestyle variables such as smoking and physical activity were retrieved from self-reported questionnaires. Smoking was used as a dichotomous variable with questions about whether participants smoke tobacco daily: yes or no.
Physical activity variables were retrieved and then constructed as a dichotomous variable: physically active or inactive (13).
Depressive symptoms
In order to measure depressive symptoms, we used the Center for Epidemiological Studies Depression Scale (CES-D).
This tool measures depressive symptoms over the preceding week, but it is not a diagnostic tool (16). CES-D contains 20 questions with response options ranging from 0 to 3 points, where 0 = ‘Rarely or none of the time’, 1 = ‘Some or a little of the time’, 2 = ‘Occasionally or a moderate amount of time’, and 3 = ‘Most or all of the time’.
The scores ranged from 0 to 60, where a high score indicates more depressive symptoms. A score of <16 indicates no sign of depressive symptoms, and ≥16 means there is a risk of depressive symptoms.
The form is validated in the USA and has been shown to be valid and reliable (16). However, it has not been validated in Norway, despite being in frequent use.
Our study found that the reliability of CES-D measured using Cronbach’s alpha had a satisfactory internal consistency with alpha = 0.77, although an alpha above 0.8 is recommended (15).
Patient education and self-management
The Health Education Impact Questionnaire (heiQ) is a questionnaire for people with chronic conditions, and consists of 40 questions (17). These questions measure health knowledge and self-management, and the questionnaire is divided into eight domains: ‘Positive and active engagement in life’, ‘Health-directed behaviour’, ‘Skill and technique acquisition’, ‘Constructive attitudes and approaches’, ‘Self-monitoring and insight’, ‘Health service navigation’, ‘Social integration and support’, and ‘Emotional well-being’.
This form has proven to be valid and reliable (17), and has been translated into Norwegian and psychometrically tested in a Norwegian sample (18).
Ethical considerations
The randomised controlled trial and associated studies were previously approved by the Regional Committees for Medical and Health Research Ethics (REC) (reference number 2010/427).
Everyone who wanted to participate in the survey received an information letter, which included a brief summary of the project and an invitation to obtain more in-depth information about participation.
More detailed information was then provided both in writing and orally, and those who wanted to participate signed an informed consent form to participate in the study (13).
Statistical analyses
The statistical analyses for this study were conducted using the analysis program SPSS, version 23. We conducted exploratory data analyses. Explanatory variables were selected based on earlier research and experience in the field.
The sociodemographic and clinical categorical variables and the treatment and lifestyle variables are presented as absolute numbers (n) and as a percentage (%).
We performed all the analyses based on available data, and did not use imputation for missing data. The continuous sociodemographic and clinical variables and the treatment and lifestyle variables are presented as a median, with minimum and maximum values (min–max).
In order to show whether there was a statistically significant association between the dichotomous variable for depressive symptoms (CES-D <16, CES-D ≥ 16) and the categorical sociodemographic and clinical variables as well as the treatment and lifestyle variables, we used Pearson’s chi-squared test.
We used the Mann-Whitney U test to establish whether there was a significant association between the dichotomous variable for depressive symptoms (CES-D <16, CES-D ≥ 16) and the continuous variables.
The variables from the univariate and unadjusted analyses that showed a statistically significant association with CES-D were included in logistic regression analyses, which were adjusted for age, gender and hypoglycaemia. The significance level chosen was 5 per cent and two-sided.
However, associations between comorbidity and CES-D scores were not analysed in our study as questions about mental health were included in the variables that measured comorbidity, which could correlate with the questions in CES-D.
We also excluded some domains in heiQ from the analyses that could correlate with CES-D, such as ‘Positive and active engagement in life’, “Health-directed behaviour’ and ‘Emotional well-being’.
Results
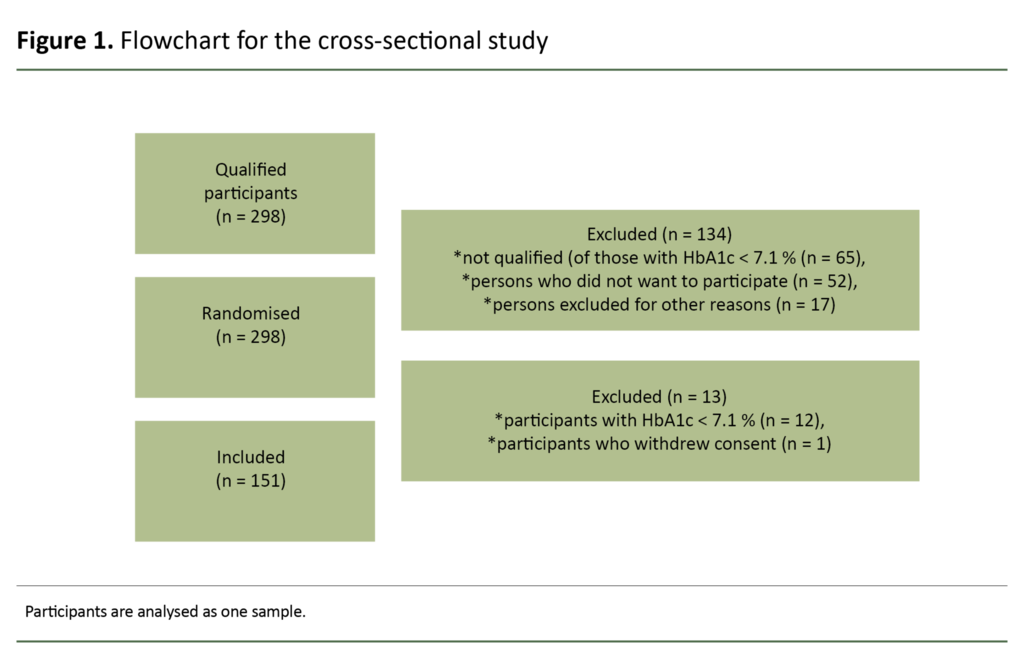
In the randomised controlled trial, a total of 298 people met the inclusion criteria, of whom 65 were excluded due to HbA1c < 7.1 per cent, 52 did not want to participate, and 17 were excluded for other reasons (Figure 1).
The inclusion criteria have already been described in detail (13). There was a total of 151 participants with type 2 diabetes at the start of the study (13). The sample in the randomised controlled trial was randomised into three groups, but in this study, they were analysed as one sample (n = 151).
Characteristics of the sample
The median age of the participants was 58 (min–max = 20–80), and 62 of the participants (41.1 per cent) were women.
Of the participants, 100 had ≤ 12 years of education (66.2 per cent), 107 (70.9 per cent) were in employment, and 41 (27.2 per cent) lived alone (Table 1). A total of 34/149 of the participants (22.5 per cent) had symptoms of depression (CES-D ≥ 16).
The median HbA1c in the participants was 7.7 per cent (min–max = 5.6–13.0) (Table 2). A total of 9 out of 131 participants (6.0 per cent) did not use glucose-lowering medication, and 142 (94.0 per cent) measured their blood sugar levels themselves.
The median body mass index (BMI) was 30.9 kg/m² (min–max = 19.9–52.6) and 98 of the participants (64.9 per cent) reported being physically inactive. The median diabetes duration was 9 years (min–max = 1–36), and 97 of the participants (64.2 per cent) had one to two concurrent illnesses (comorbidity) in addition to type 2 diabetes.
The only statistically significant association we found between CES-D scores and participant characteristics was for employment status (Table 1) and hypoglycaemia (Table 2).
Unadjusted analyses
Table 3 shows self-management as measured by the heiQ and the association with reported depressive symptoms (CES-D). We found that those who reported depressive symptoms scored lower on six out of eight self-management domains measured by the heiQ.
Adjusted analyses
Logistic regression showed an association between younger participants and an increased likelihood of reporting depressive symptoms compared to non-depressive symptoms (p = 0.042), adjusted for gender and hypoglycaemia.
Furthermore, we found that the odds of reporting hypoglycaemia were higher among those who reported depressive symptoms than those without depressive symptoms (p = 0.021), adjusted for age and gender (Table 4).
Logistic regression showed an association between younger participants and an increased likelihood of reporting depressive symptom.
Analyses of the participants’ employment status showed that those who reported depressive symptoms were eight times more likely to be unemployed than those without depressive symptoms, adjusted for age and gender.
Analyses of self-management (heiQ) showed that those who reported depressive symptoms were 5 per cent more likely to have a low score on ‘Constructive attitudes and approaches’ than those without depressive symptoms (p < 0.001).
We also found that those reporting depressive symptoms were more likely to report a low score on ‘Skill and technique acquisition’ and ‘Social integration and support’ – 19 and 25 per cent respectively.
Discussion
We found that as many as one-fifth of participants (22.5 per cent) in the Norwegian randomised controlled trial reported symptoms of depression (CES-D ≥ 16). Younger participants were more likely to report depressive symptoms.
Those with lower scores on the heiQ domain ‘Social integration and support’ were more likely to report depressive symptoms.
Those who reported depressive symptoms were also eight times more likely to be out of work.
As many as one-fifth of participants in the Norwegian randomised controlled trial reported symptoms of depression.
Furthermore, there was an increased likelihood of a lower score on the heiQ domain ‘Constructive attitudes and approaches’ and the scores were lower on ‘Skill and technique acquisition’ among those reporting depressive symptoms.
It was also found that those who reported experiencing hypoglycaemia were more likely to report depressive symptoms.
One-fifth reported symptoms of depression
We found that about one-fifth of participants reported symptoms of depression.
However, earlier research in Norway into healthy older people using the same measuring instrument (CES-D) has shown that more than one in five people over the age of 75 have symptoms of depression, and that along with loneliness, this symptom appears more frequently in the youngest and oldest groups than in those in the intermediate stage of life (19).
This finding is consistent with the findings in our study, where younger participants were more likely to report depressive symptoms.
The American Diabetes Association (ADA) recommends screening all older adults (aged ≥ 65 years) with diabetes for cognitive impairment and depression (9), and has previously listed a number of possible assessment tools (20).
Earlier research has also shown that younger patients may be more open to discussing psychosocial needs than patients over the age of 65, and that many patients are unaware that psychosocial well-being factors can also be addressed in a consultation (21).
Lack of social integration associated with depressive symptoms
A lower score in the heiQ domain ‘Social integration and support’ was found in this sample to be associated with an increased likelihood of reporting depressive symptoms.
It has been shown previously that lack of social support can be a risk factor for increased mortality, and that a person’s social support should be taken just as seriously as other risk factors, such as smoking and hypertension (22).
In our sample, those who reported depressive symptoms were eight times more likely to be out of work than those who did not report depressive symptoms.
It has been shown previously that lack of social support can be a risk factor for increased mortality.
Recent research shows that people who are out of work can benefit from support, especially when it is from someone in the same situation as themselves. Research also shows that those with a poorer health potential find their situation more challenging than those who are healthy (23).
Constructive reasoning reduced the risk of depressive symptoms
We also found that participants with a higher score on ‘Constructive attitudes and approaches’ in relation to managing their diabetes were less likely to have symptoms of depression.
It has also been shown that a person’s attitude to the treatment of the condition impacts on how they exercise self-management (24). However, it is not only the patient’s attitude to their diabetes management that determines the outcome, but also the attitudes of the healthcare personnel.
Time constraints and staff shortages in the health service can also represent a barrier to developing and implementing patient education, good treatment plans and medication follow-up (24).
In Norway, only 45 per cent of those with type 2 diabetes reach their HbA1c target level (25). This may be partly due to the fact that many patients with type 2 diabetes struggle to follow the treatment regimen (24, 25).
In our study, we found no statistically significant association between HbA1c and incidence of symptoms of depression, and this finding is consistent with earlier research (26).
Skills and knowledge of assistive tools have a positive effect
However, our findings show that a high score on ‘Skill and technique acquisition’ may reduce the likelihood of people with type 2 diabetes having symptoms of depression, which may be due to the person having good skills and knowledge of assistive tools to protect their own health and facilitate diabetes management (17).
Patients with hyperglycaemia are often given glucose-lowering medication, which can increase the risk of hypoglycaemia. Those who reported experiencing hypoglycaemia in our study were more likely to report depressive symptoms, which is consistent with earlier research (27).
Identifying the patient’s emotional concerns about their diabetes is therefore an important part of diabetes management for this patient group (27).
Healthcare personnel must be able to recognise the signs of depression
While the IDF suggests that people with diabetes should be screened for complications such as depression (1, 11), this is not recommended in Norway as there is limited evidence that such an intervention has any effect (12).
Instead, Norwegian national clinical guidelines for diabetes suggest screening for depression if there is a clinical suspicion of such, and stress that healthcare personnel must be observant of the increased occurrence of depression in people with diabetes (12).
Symptoms of depression in those with type 2 diabetes can be just as widespread and even more common than diagnosed severe depression, and earlier research has shown that those with symptoms of depression are not followed up as closely as those who have been diagnosed with depression (26).
Symptoms of depression in those with type 2 diabetes can be just as widespread and even more common than diagnosed severe depression.
In the randomised controlled trial, where we tested a diabetes diary mobile app, we did not find any effect of the app on the primary endpoint of HbA1c, and we have previously discussed a number of possible reasons for this (13–15).
Depression may be one reason, and as a consequence of an absence of self-management, the participants may have needed a higher intensity intervention than the app they were given.
Research shows that screening tools such as patient-reported outcome measures (PROMs) can also be used to promote communication between the patient and healthcare personnel, enabling the patient to become more involved with their own treatment (28).
The term ‘patient-reported outcome’ (PRO) is defined as ‘any report of the patient’s health condition that comes directly from the patient, without interpretation of the patient’s response by a clinician or anyone else’ (29).
Research shows that using PROM does not take up more time in the clinical consultation (29), which is an important point since healthcare personnel face many demands on their time (24).
Tools for screening for psychosocial factors are already being used in Norway (30), and the findings in our study show that more research is needed into whether high-risk people with diabetes should be screened for depression in the same way that they are monitored for HbA1c in regular check-ups.
Strengths and weaknesses of the study
In this study, we used the heiQ questionnaire, which is divided into eight domains. We did not use all of these domains, as some of them, such as ‘Emotional well-being’, measured similar phenomena as those measured in CES-D.
Likewise, we did not investigate the association between comorbidity and CES-D since mental health was covered in comorbidity and in CES-D, and the measurements could therefore be correlated.
The three selected domains from heiQ in this article clearly illustrate how poorer self-management is associated with depressive symptoms.
The fact that the CES-D questionnaire is validated internationally (16) but not psychometrically tested in Norway is a weakness of the study. The results of this study therefore entail a degree of uncertainty.
On the other hand, the number reporting depressive symptoms is consistent with the Norwegian study on life course, ageing and generation (NorLAG), which uses the same questionnaire (19).
The sample in this study had volunteered to participate in a randomised controlled trial for the purpose of changing their lifestyle using a mobile app for self-management. It is therefore a further weakness of this study that those recruited may have been extra motivated to change behaviour, and as such, not representative of people with type 2 diabetes.
However, it emerged that many of the participants found it difficult to change their lifestyle (31). Nevertheless, the method of recruitment is a limitation in a study like this.
Another methodological limitation is that our investigation was exploratory, with a view to identifying associations. The findings need to be confirmed in future research with a larger sample, and using other methods where appropriate.
The study was cross-sectional, which means we are not able to explain any causal relationship, and our intention was only to examine associations.
Conclusion
We found that one-fifth of participants with type 2 diabetes in a randomised controlled trial with a mobile self-management diabetes diary app reported symptoms of depression upon inclusion in the trial.
The patients who reported depressive symptoms generally scored lower on self-management (heiQ). We found that depressive symptoms were more frequent in younger people.
Those with depressive symptoms may need more support from nurses and other healthcare personnel to manage their condition.
It has therefore been discussed whether screening for depressive symptoms in high-risk people with diabetes is appropriate, both in Norway and internationally. This issue is relevant to research and clinical practice.
References
1. International Diabetes Federation. International Diabetes Federation Diabetes Atlas; 2017. 8th ed. Available at: http://www.diabetesatlas.org/ (downloaded 14.08.2018).
2. Stene LC, Strøm H, Gulseth HL. Diabetes i Norge. Oslo: Folkehelseinstituttet; 2014. Available at: https://www.fhi.no/nettpub/hin/helse-og-sykdom/diabetes-i-norge---folkehelserappor/ (downloaded 14.08.2018).
3. Holt R, de Groot M, Golden S. Diabetes and depression. Current Diabetes Reports. 2014;14(6):1–9.
4. Rotella F, Mannucci E. Diabetes mellitus as a risk factor for depression. A meta-analysis of longitudinal studies. Diabetes Research and Clinical Practice. 2013;99(2):98–104.
5. Egede LE. Diabetes, major depression, and functional disability among U.S. adults. Diabetes Care. 2004;27:421–8.
6. Park M, Katon WJ, Wolf FM. Depression and risk of mortality in individuals with diabetes: a meta-analysis and systematic review. General Hospital Psychiatry. 2013;35(3):217–25.
7. Nouwen A, Winkley K, Twisk J, Lloyd C, Peyrot M, Ismail K, et al. Type 2 diabetes mellitus as a risk factor for the onset of depression: a systematic review and meta-analysis. Clinical and Experimental Diabetes and Metabolism. 2010;53(12):2480–6.
8. Barlow J, Wright C, Sheasby J, Turner A, Hainsworth J. Self-management approaches for people with chronic conditions: a review. Patient Education and Counseling. 2002;48(2):177–87.
9. American Diabetes Association. 5. Lifestyle management. Standards of medical care in diabetes 2019. Diabetes Care. 2019;42(Suppl. 1):S46–60. DOI: 10.2337/dc19-S005
10. Richards D, Chellingsworth M, Hope R, Turpin G, Whyte M. Reach out: national programme supervisor materials to support the delivery of training for psychological wellbeing practitioners delivering low intensity interventions 2010. Available at: http://www.babcp.com/files/Accreditation/PWP/IAPT-PWP-Supervision-Manual-Reach-Out.pdf (downloaded 14.08.2018).
11. International Diabetes Federation. Recommendations For Managing Type 2 Diabetes In Primary Care. Brussel; 2017. Available at: www.idf.org/managing-type2-diabetes (downloaded 14.08.2018).
12. Helsedirektoratet. Nasjonal faglig retningslinje for diabetes. Oslo; 2017. Available at: https://helsedirektoratet.no/retningslinjer/diabetes (downloaded 14.08.2018).
13. Torbjørnsen A, Jenum AK, Småstuen MC, Årsand E, Holmen H, Wahl AK, Ribu L. A low-intensity mobile health intervention with and without health counseling for persons with type 2 diabetes, part 1: baseline and short-term results from a randomized controlled trial in the Norwegian part of RENEWING HEALTH. JMIR mHealth and uHealth. 2014;2(4):e52.
14. Holmen H, Torbjørnsen A, Wahl AK, Jenum AK, Småstuen MC, Årsand E, Ribu L. A mobile health intervention for self-management and lifestyle change for persons with type 2 diabetes, part 2: one-year results from the Norwegian randomized controlled trial RENEWING HEALTH. JMIR mHealth and uHealth. 2014;2(4):e57.
15. Holmen H. Mobile health for diabetes self-management. (Doctoral thesis.) Oslo: Universitetet i Oslo, Medisinsk fakultet; 2017.
16. Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Applied Psychological Measurement. 1977 juni;1(3):385–401.
17. Osborne RH, Elsworth GR, Whitfield K. The Health Education Impact Questionnaire (heiQ): An outcomes and evaluation measure for patient education and self-management interventions for people with chronic conditions. Patient Education and Counseling. 2007;66(2):192–201.
18. Wahl AK, Osborne RH, Langeland E, Wentzel-Larsen T, Mengshoel AM, Ribu L, et al. Making robust decisions about the impact of health education programs: Psychometric evaluation of the Health Education Impact Questionnaire (heiQ) in diverse patient groups in Norway. Patient Education and Counseling. 2016;99(10):1733.
19. Hansen T, Daatland SO. Aldring, mestringsbetingelser og livskvalitet. Oslo: Helsedirektoratet; 2016.
20. Young-Hyman D, de Groot M, Hill-Briggs F, Gonzalez JS, Hood K, Peyrot M. Psychosocial care for people with diabetes: A position statement of the American Diabetes Association. Diabetes Care. 2016;39(12), 2126–40.
21. van Dijk-de Vries A, van Bokhoven MA, de Jong S, Metsemakers JFM, Verhaak PFM, van Der Weijden T, et al. Patients’ readiness to receive psychosocial care during nurse-led routine diabetes consultations in primary care: a mixed methods study. International Journal of Nursing Studies. 2016;63:58–64.
22. Holt-Lunstad J, Smith TB, Layton JB, Brayne C. Social relationships and mortality risk: a meta-analytic review (social relationships and mortality). PLoS Medicine. 2010;7(7):e1000316.
23. Heggebø K, Elstad JI. Is it easier to be unemployed when the experience is more widely shared? Effects of unemployment on self-rated health in 25 European countries with diverging macroeconomic conditions. European Sociological Review. 2018;34(1):22–39.
24. Nam S, Chesla C, Stotts NA, Kroon L, Janson SL. Barriers to diabetes management: Patient and provider factors. Diabetes Research and Clinical Practice. 2011;93(1):1–9.
25. Mouland G. Diabetes i en fastlegepraksis – ble behandlingsmålene nådd? Tidsskrift for Den norske legeforening. 2014;134(2):168–72.
26. Golden SH, Shah N, Naqibuddin M, Payne JL, Hill-Briggs F, Wand GS, et al. The prevalence and specificity of depression diagnosis in a clinic-based population of adults with type 2 diabetes mellitus. Psychosomatics. 2017;58(1):28–37.
27. Pearce MJ, Pereira K, Davis E. The psychological impact of diabetes: a practical guide for the nurse practitioner. Journal of the American Association of Nurse Practitioners. 2013;25(11):578–83.
28. Snyder CF, Aaronson NK, Choucair AK, Elliott TE, Greenhalgh J, Halyard MY, et al. Implementing patient-reported outcomes assessment in clinical practice: a review of the options and considerations. Quality of Life Research. 2012;21(8):1305–14.
29. Santana MJ, Haverman L, Absolom K, Takeuchi E, Feeny D, Grootenhuis M, et al. Training clinicians in how to use patient-reported outcome measures in routine clinical practice. Quality of Life Research. 2015;24(7):1707–18.
30. Hernar I. Bruk av pasientrapporterte målinger for å bedre kvaliteten på oppfølgingen av personer med diabetes – en feasibility studie. Bergen: Helse Bergen HF, Haukeland universitetssjukehus; 2017. Available at: https://helseforskning.etikkom.no/prosjekterirek/prosjektregister/prosjekt?p_document_id=796318&p_parent_id=802344&_ikbLanguageCode=us (downloaded 14.08.2018).
31. Ribu L, Rønnevig M, Corbin J. People with type 2 diabetes struggling for self-management: A part study from the randomised controlled trial in RENEWING HEALTH. Nursing Open. 23.05.2019;1–9.














Comments